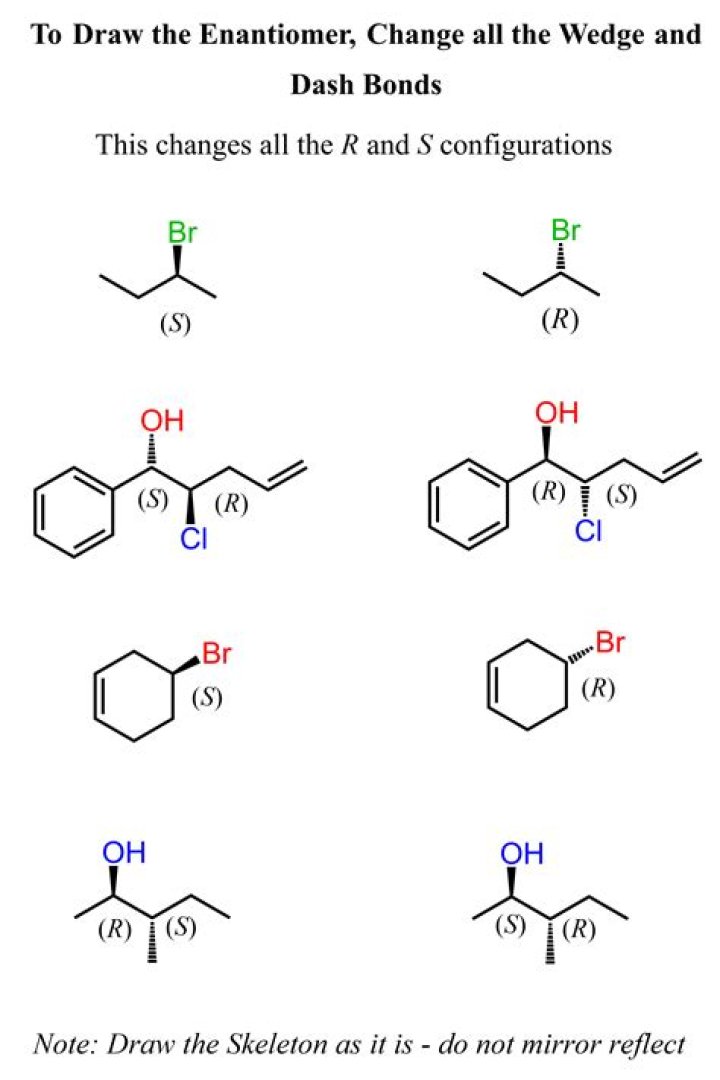
The R / S system is an important nomenclature system for denoting enantiomers. This approach labels each chiral center R or S according to a system by which its substituents are each assigned a priority, according to the Cahn–Ingold–Prelog priority rules (CIP), based on atomic number..
In this way, what is the difference between R and S enantiomers?
Because the 4th highest priority atom is placed in the back, the arrow should appear like it is going across the face of a clock. If it is going clockwise, then it is an R-enantiomer; If it is going counterclockwise, it is an S-enantiomer.
Also, is r and s the same as L and D? R and S refer to the structural difference between stereoisomers. D and L d and l can only be measured by determining whether the substance rotates the polarization of polarized light clockwise or counterclockwise.
Regarding this, what is r and s in chemistry?
R and S Notation[edit] Follow the direction of the remaining 3 priorities from highest to lowest priority (lowest to highest number, 1<2<3). A counterclockwise direction is an S (sinister, Latin for left) configuration. A clockwise direction is an R (rectus, Latin for right) configuration.
Are D and L enantiomers?
D and L enantiomers[edit] D and L enantiomers refer to the configurational stereochemistry of the molecule. L isomers have the hydroxy group attached to the left side of the asymmetric carbon furthest from the carbonyl, while D isomers have the hydroxy group on the right side.
Related Question Answers
Are enantiomers always chiral?
Enantiomers are always chiral, but diastereomers may or may not be chiral.What does R and S mean in stereochemistry?
The Cahn-Ingold-Prelog system is a set of rules that allows us to unambiguously define the stereochemical configuration of any stereocenter, using the designations 'R ' (from the Latin rectus, meaning right-handed) or ' S ' (from the Latin sinister, meaning left-handed).What is D and L?
The D,L convention, not to be confused with the d and l descriptors used to designate the direction of specific rotation of chiral compounds, is a convention used to distinguish between enantiomers of chiral monosaccharides and chiral alpha-amino acids, based on the molecule drawn as a Fischer projection in a specificAre diastereomers chiral?
Diastereomers are always chiral, and always different from one another. Note that diasteriomers occur in pairs, and each has two chiral centers.What is S and R configuration?
The R / S system is an important nomenclature system for denoting enantiomers. This approach labels each chiral center R or S according to a system by which its substituents are each assigned a priority, according to the Cahn–Ingold–Prelog priority rules (CIP), based on atomic number.What is D in organic chemistry?
Deuterium is frequently represented by the chemical symbol D. Since it is an isotope of hydrogen with mass number 2, it is also represented by 2. H. .What is the relationship between R S and D L?
(D-L system labels the whole molecule, while R/S system labels the absolute configuration of each chirality center.) In short, the D-L system doesn't have direct connection to (+)/(-) notation. It only relates the stereochemistry of the compound with that of glyceraldehyde, but says nothing about its optical activity.How do I assign priority to R and S?
Assign the priority (high = 1 to low = 4) to each group attached to the chirality center based on atomic number. Reposition the molecule so that the lowest priority group is away from you as if you were looking along the C-(4) σ bond. If you are using a model, grasp the lowest priority group in your fist.How do you know if a chiral center is R or S?
Draw a curve from the first-priority substituent through the second-priority substituent and then through the third. If the curve goes clockwise, the chiral center is designated R; if the curve goes counterclockwise, the chiral center is designated S.What does it mean to be chiral?
Definitions: Chiral. A molecule is chiral if it is not superimposable on its mirror image. Most chiral molecules can be identified by their lack of a plane of symmetry or a center of symmetry. Your hand is a chiral object, as it does not have either of these types of symmetry.What is the difference between enantiomers and diastereomers?
There are two types of stereoisomers— enantiomers and diastereomers. Enantiomers contain chiral centers that are mirror images and non-superimposable. Diastereomers contain chiral centers that are non-superimposable but are NOT mirror images. There can be many more than 2 depending on the number of stereocenters.What does E and Z mean in organic chemistry?
E comes from the German entgegen which means opposite. So the two isomers are: Summary. (E)- : the higher priority groups are on opposite sides of the double bond. (Z)- : the higher priority groups are on the same side of the double bond.How many stereoisomers are possible for?
Three stereoisomers are possible: one pair of enantiomers (A and B) and an achiral molecule C, called a “meso compound.” A meso compound is an achiral molecule that nonetheless contains a stereogenic atom.What is r prime in organic chemistry?
In chemistry, it is used to distinguish between different functional groups connected to an atom in a molecule, such as R and R′, representing different alkyl groups in an organic compound. In molecular biology, the prime is used to denote the positions of carbon on a ring of deoxyribose or ribose.What does R stand for in pV nRT?
The ideal gas law is: pV = nRT, where n is the number of moles, and R is universal gas constant. The value of R depends on the units involved, but is usually stated with S.I. units as: R = 8.314 J/mol·K.Are hands chiral or achiral?
When the thumbs point in the same direction, the palms are opposite. Your hands are mirror images, but not superposable. Each hand is therefore chiral. Achiral objects may be superposed on their mirror image.What is a chiral atom?
An asymmetric carbon atom (chiral carbon) is a carbon atom that is attached to four different types of atoms or groups of atoms. Molecules that cannot be superimposed on their own mirror image are said to be chiral like mirror image.How do you find chiral centers?
Chiral molecules usually contain at least one carbon atom with four nonidentical substituents. Such a carbon atom is called a chiral center (or sometimes a stereogenic center), using organic-speak. Any molecule that contains a chiral center will be chiral (with the exception of a meso compound).What is D and L form of sugar?
For a sugar drawn in the Fischer projection with the most oxidized carbon at the top (i.e. aldehyde or ketone) if the OH on the bottom chiral centre points to the right, it is referred to as D- if the OH on the bottom chiral centre points to the left, it is referred to as L- .