Yes. A molecule does exist that is composed of two oxygen atoms and one hydrogen atom bonded together. The substance is called hydroperoxyl radical and can be represented as HO2. Covalent bonds hold the two oxygen atoms and one hydrogen atom together..
Moreover, why there are two hydrogen atoms for each oxygen atom?
When two hydrogen atoms come close enough to an oxygen atoms, their electrons are attracted to the proton of the other atom. Because there is both a strong enough attraction between atoms and room for electrons in the outer energy levels of the atoms, they share electrons.
Secondly, what is two oxygen and hydrogen? Hydrogen Peroxide Two hydrogen (H) atoms can also bond with two oxygen (O) atoms, making the formula H2O2. That's just one more oxygen than water, but it is a totally different compound. You can see that each of the Oxygen atoms has eight electrons, and the two Hydrogens have two electrons each.
Similarly one may ask, how do you combine hydrogen and oxygen?
To combine hydrogen and oxygen to make water, you basically have to mix the gases together and light them with a match. Just mixing the gases together isn't enough - you have to do something to get the chemical reaction started. The problem is that this creates a big explosion.
What are 2 hydrogen atoms called?
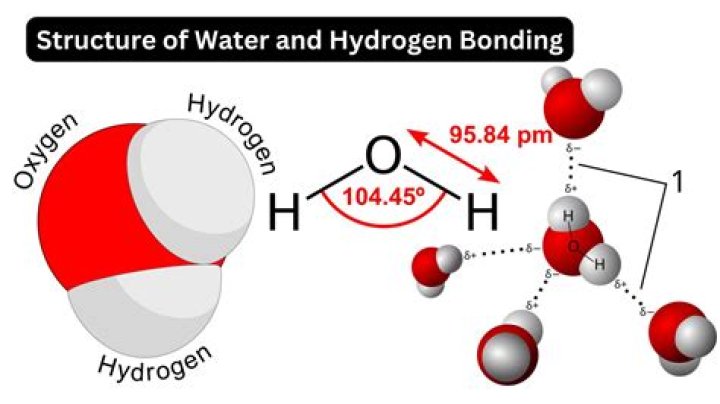
Water is made up of two hydrogen (H) atoms and one oxygen (O) atom. The formula for water is H2O.
Related Question Answers
What type of bond is water?
Water is a polar molecule A water molecule is formed when two atoms of hydrogen bond covalently with an atom of oxygen. In a covalent bond electrons are shared between atoms. In water the sharing is not equal. The oxygen atom attracts the electrons more strongly than the hydrogen.How many atoms are in h2o?
For H2O, there is one atom of oxygen and two atoms of hydrogen.What is Oxygen's atom called?
Oxygen is a chemical element – a substance that contains only one type of atom. Its official chemical symbol is O, and its atomic number is 8, which means that an oxygen atom has eight protons in its nucleus. Oxygen is normally found as a molecule. It is called dioxygen.What type of bond is NaCl?
Ionic compounds, such as sodium chloride (NaCl), are formed by a transfer of electrons that creates ions. Ions exert electrostatic force on each other, which forms ionic bonds. The hydrogen and oxygen atoms in a water molecule, however, are bonded by sharing electrons rather than by transferring them.How many atoms does nitrogen have?
Under standard conditions nitrogen is a colorless, tasteless, odorless gas. It forms diatomic molecules, which means that there are two nitrogen atoms per molecule in nitrogen gas (N2).How many electrons does oxygen atom have?
2, 6
How many atoms are in a gram?
To calculate the number of atoms in a sample, divide its weight in grams by the amu atomic mass from the periodic table, then multiply the result by Avogadro's number: 6.02 x 10^23.What happens if you mix hydrogen and oxygen?
Hydrogen molecules violently react with oxygen when the existing molecular bonds break and new bonds are formed between oxygen and hydrogen atoms. As the products of the reaction are at a lower energy level than the reactants, the result is an explosive release of energy and the production of water.Can oxygen and hydrogen form a covalent bond?
The covalent bonds between hydrogen and oxygen atoms in water are polar covalent bonds. The shared electrons spend more time near the oxygen nucleus, giving it a small negative charge, than they spend near the hydrogen nuclei, giving these molecules a small positive charge.How many atoms of oxygen are in 2h2o?
A water molecule (formally known as dihydrogen monoxide) is composed of two hydrogen atoms and one oxygen atom. But you can't simply take two hydrogen atoms and stick them onto an oxygen atom. The actual reaction to make water is a bit more complicated: 2H2 + O2 = 2H2O + Energy.Can you combine oxygen and hydrogen to make water?
To combine hydrogen and oxygen to make water, you basically have to mix the gases together and light them with a match. Just mixing the gases together isn't enough - you have to do something to get the chemical reaction started. This reaction will produce lots and lots of bubbles (carbon dioxide) as well as water.What type of reaction is hydrogen and oxygen?
Exothermic Reaction After one set of hydrogen and oxygen molecules have reacted, the energy released triggers molecules in the surrounding mixture to react, releasing more energy. The result is an explosive, rapid reaction that releases energy quickly in the form of heat, light and sound.What elements does oxygen react with?
Oxygen is very reactive with Alkali metals. Alkali metals are given the name alkali because the oxides of these metals react with water to form a metal hydroxide that is basic or alkaline. Lithium produces an oxide, sodium produces a peroxide, and potassium, cesium, and rubidium produce superoxides.Can we create water?
Theoretically, this is possible, but it would be an extremely dangerous process, too. To create water, oxygen and hydrogen atoms must be present. We've got water. The hydrogen and oxygen atoms' electrons' orbits have been conjoined.Is hydrogen and oxygen a compound?
All compounds are molecules, but not all molecules are compounds. Hydrogen gas (H2) is a molecule, but not a compound because it is made of only one element. Water (H2O) can be called a molecule or a compound because it is made of hydrogen (H) and oxygen (O) atoms.How do you make pure oxygen?
To make oxygen in the laboratory, hydrogen peroxide is poured into a conical flask containing some manganese(IV) oxide. The gas produced is collected in an upside-down gas jar filled with water. As the oxygen collects in the top of the gas jar, it pushes the water out.How many atoms does kno3?
Each nitrate ion contains one nitrogen atom and three oxygen atoms.How many atoms are in Sulfur?
Expert Answer 1 mole of any substance consists of 6.022 x 1023 atoms of that substance. Now since 1 mol of sulfur has 6.022 x 1023 atoms of sulfur, Therefore, 3.How many atoms are in h2so4?
One sulfuric (H2SO4) molecule has 2 hydrogen atoms, 1 sulfur atom, and 4 oxygen atoms. You can also say one mole of sulfuric acid has two mols of hydrogen atoms, 1 mol of sulfur atoms, and 4 moles of oxygen atoms. So in total, we have 7 mols of atoms. But that's in 1 mol of sulfuric acid.