.
Likewise, people ask, why will Aluminium not react with potassium nitrate?
When fine aluminum and potassium nitrate are in the same comp and when it is dampened with water, it has the potential for a run away exothermic reaction which can end in the composition igniting. Pumped stars contain much less water and are not as suceptible to this reaction. Many pyros never see this reaction occur.
Additionally, what happens when you mix aluminum and iron oxide? A lot of heat is needed to start the reaction, but then it releases an incredible amount of heat, enough to melt the iron. Because aluminium is more reactive than iron, it displaces iron from iron(III) oxide. The aluminium removes oxygen from the iron(III) oxide: iron is reduced.
Also know, what happens when you mix oxygen and potassium?
Considerable energy is released, and potassium oxide is formed, as the potassium burns. The reaction equation is 4K + O2 → 2K2O. The flame in this reaction has a violet color.
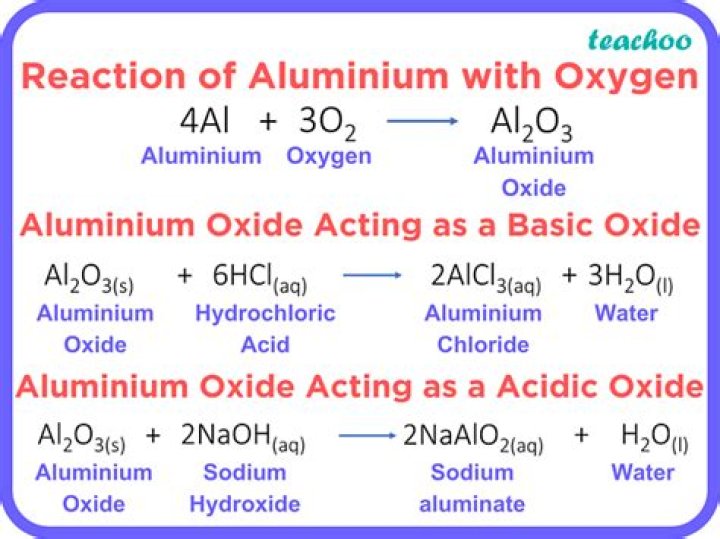
Does Aluminium oxide and carbon react?
Aluminum oxide react with carbon (coke) to produce aluminum carbide and carbon monoxide. This reaction takes place at a temperature near 1800°C.
Related Question AnswersCan you use aluminum oxide in thermite?
Is it possible to use aluminum oxide instead powdered aluminum? No. Thermite is an oxidation of the aluminum powder to aluminum oxide and a concurrent reduction of the iron oxide powder to iron. The aluminum and iron oxidation and reduction, respectively, form the two half cells of the thermite redox reaction.Why is there no reaction between Aluminium and silver nitrate?
Aluminum metal in silver nitrate …. The passivating layer of Al2O3 will prevent aluminum from reducing silver ions. Al(s) + AgNO3(aq) → No reaction. The two things we might do to the mixture to disrupt the Al2O3 surface will cause the Ag+ ions to react.Which is the most reactive metal?
cesiumIs magnesium more reactive than iron?
Aluminium is much higher than iron in the reactivity series, so the thermite reaction releases a lot of energy. Magnesium is very high in the reactivity series, and copper is very low – so the reaction between magnesium and copper(II) oxide is more violent.Why is magnesium more reactive than iron?
Why is magnesium so reactive, compared to other elements like zinc and aluminum? Because magnesium loses its electrons easily than zinc and aluminum, other elements and compounds like oxygen, water and the halogens reacts vigorously with atoms that loses electron easily.Can Aluminium displace copper?
Aluminium appears less reactive than copper. The aluminium foil appears unable to displace copper from copper(II) sulfate solution. 3. Scratches on the surface of the oxide layer allow chloride ions to react with aluminium, this effects the cohesiveness of the oxide layer.Where is aluminum in the reactivity series?
| Substance | Comment |
|---|---|
| Sodium | Metal: very reactive |
| Calcium | Metal: very reactive |
| Magnesium | Metal: fairly reactive |
| Aluminium | Metal: fairly reactive |
Does Iron and Aluminium oxide react?
Aluminium reacts with iron(III) oxide because it can be considered to be more reactive than iron so will remove oxygen from the iron(III) oxide.What two elements react violently together?
Reactivity series of metals| Order of reactivity | Metal | Reactions with water or steam |
|---|---|---|
| most reactive | potassium (K) | very vigorous reaction with cold water |
| ↑ | sodium (Na) | vigorous reaction with cold water |
| ↓ | calcium (Ca) | less vigorous reaction with cold water |
| least reactive | magnesium (Mg) | slow reaction with cold water, vigorous with steam |