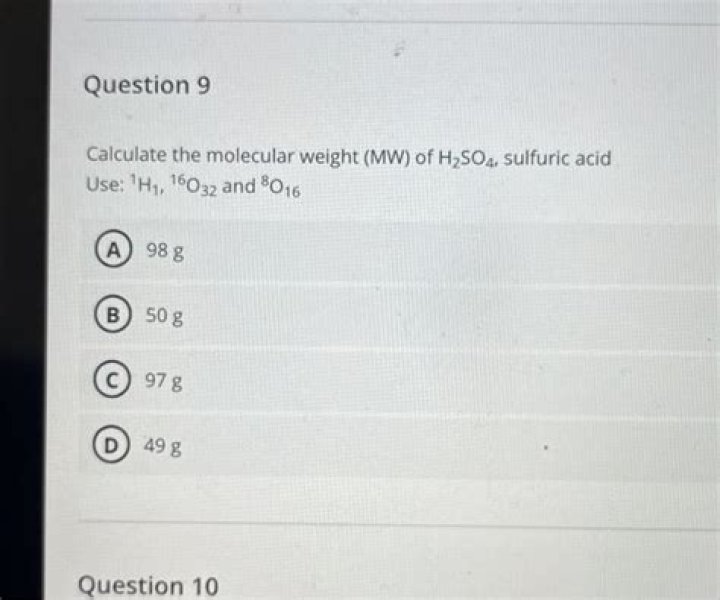
Molar mass of H2SO4 can be calculated byaddingthe respective molar masses of all the elementsconstitutingit. Molar mass of H(x2)+Molar mass ofSulphur(x1)+Molar mass of Oxygen(x4). =>98g/mol..
Subsequently, one may also ask, what is the molecular weight of h2so4?
98.079 g/mol
Beside above, how do I calculate molecular weight? Calculate the total mass for each element inthemolecule. Multiply the atomic mass of each element by thenumberof atoms of that element: (Atomic Mass of Element) x (# ofatoms ofthat element). Do this for each element in themolecule. Inour carbon dioxide example, the mass of thesingle carbon atom is12.011 amu.
Similarly, it is asked, how do you find the equivalent weight of h2so4?
Equivalent weights may be calculated frommolarmasses if the chemistry of the substance is well known:sulfuricacid has a molar mass of 98.078(5)gmol−1, and supplies two molesofhydrogen ions per mole of sulfuric acid, soitsequivalent weight is 98.078(5)gmol−1/2eqmol−1 = 49.039(3)geq−1.
What is the percentage oxygen by weight in h2so4?
Percent composition by element
| Element | Symbol | Mass Percent |
| Hydrogen | H | 2.055% |
| Oxygen | O | 65.251% |
| Sulfur | S | 32.693% |
Related Question Answers
What is normality formula?
It is defined as the gram equivalent weight per literofsolution. Here, a more favorable calculationfornormality is considered, where the normality ofasolution is equal to the molarity multiplied by the numberofequivalents. Normality(N) = Molarity(M) x numberofequivalents. Normality Formula Questions: 1.What is molecular mass in chemistry?
Molecular Mass Definition Molecular mass is a number equal to the sumofthe atomic masses of the atoms in a molecule.Themolecular mass gives the mass of amoleculerelative to that of the 12C atom, whichis taken to havea mass of 12.What is equivalent weight formula?
Equivalent weight (EW) is the molar massofa substance divided by the n number of equivalents in thesubstance.For acids or bases, the n number of equivalents is thenumber ofhydroxide ions in the base (OH-1) , and the number ofhydrogen ionsin the acid (H+1).What is meant by normality?
Normality is a measure of concentration equaltothe gram equivalent weight per liter of solution. Gramequivalentweight is the measure of the reactive capacity of amolecule. Thesolute's role in the reaction determines thesolution'snormality. Normality is also known as theequivalentconcentration of a solution.Is molecular weight the same as molar mass?
Molar mass and molecular weight areoftenconfused, but their values are very different. Molarmass isthe mass of one mole of a substance, whilemolecularweight is the mass of one molecule ofasubstance. One mole is the number of particles, such asatoms,molecules, ions or electrons, in asubstance.How many molecules are in a mole?
Chemists generally use the mole as the unitforthe number of atoms or molecules of a material.Onemole (abbreviated mol) is equalto6.022×1023 molecular entities (Avogadro'snumber),and each element has a different molar mass depending onthe weightof 6.022×1023 of its atoms(1mole).What does N mean in chemistry concentration?
(1) Normal or normal concentration refers toaconcentration of solutes that is the same intwosamples. (2) Normality is the gram equivalent weight ofasolution in a solution, which is its molarconcentrationdivided by an equivalence factor. Normalconcentration isalso known as normality, N,isotonic.What is the equivalent weight of HCL?
Examples. 1) The gram-equivalent weight of HClisone mole of HCl (1.008 + 35.453 = 36.461 g). 2)Thegram-equivalent weight of H2SO4isusually ½ mole since both hydrogens are replaceable inmostreactions of sulfuric acid.What is equivalent weight of NaOH?
The equivalent mass of NaOH is 40 grams. Itisaccording to the formula , Gram molecular weight dividedbythe 'n' factor.How do you find the equivalent weight of NaCl?
Therefore, the equivalent of NaCl willbeequal to equivalent weight of Na + equivalentweightof Cl or, 23+35.5 grams. So, we can firstcalculate theequivalent weight of each element in thecompound and thenadd them up.What is the equivalent weight of CaCO3?
Molar mass (CaCO3) = M(Ca) + M(C) + 3xM(O) = 40 + 12 + 3 x 16 = 100 g/mole. Divide the molarmassby the ion charge or oxidation number (for CaCO3)todetermine equivalent (Eq.) weights. Eq.weight(HCO3(-)) = 61 / 1 (charge) = 61 g/Eq.What is equivalent weight of kmno4?
The molar mass of potassium permanganate is158g/mol. To find the equivalent mass, divide thatmolarmass by the mols of electrons taken in its halfreaction.That would be five. 158/5 =31.6grams/equivalent.What is the formula for calculating molecular weight?
Sample Molecular Weight Calculation The chemical formula for water is H2O,whichmeans this molecule has 3 atoms: 2 of hydrogen (H) and1oxygen (O) atom. In order to calculate themolecularweight of one water molecule, we add thecontributionsfrom each atom; that is, 2(1) + 1(16) =18grams/mole.How do you find the molecular weight of NaOH?
The molar mass of sodium hydroxide equals39.997g/mol. To determine the molar mass,multiplythe atomic mass by the number of atoms intheformula.What is the difference between molecular weight and formula weight?
The formula mass (formula weight) ofamolecule is the sum of the atomic weights oftheatoms in its empirical formula. The molecularmass(molecular weight) of a molecule is its averagemassas calculated by adding together the atomic weights oftheatoms in the molecular formula.How many moles are in a gram?
Amazingly, there are 6.02x10^23 atoms in each ofthesamples above. A sample of 12 grams of carbon is equaltoone mole. The amount of moles in a substance canbedetermined using that substance's molar mass. The molar mass istheamount of grams in one mole ofasubstance.What is h2so4 made up of?
Sulfuric acid (alternative spellingsulphuricacid), also known as vitriol, is a mineral acidcomposed ofthe elements sulfur, oxygen and hydrogen, withmolecular formulaH2SO4.What is the formula of h2so4?
Sulfuric Acid (H2SO4) is a strongmineralacid that has is colorless when pure. This chemical is usedas achemical intermediate to manufacture other chemicals andcleaningmetal surfaces. The formula for sulfuric acidisH2SO4. The molar mass of sulfuric acid is 98.07848gmol.