.
Besides, how do you identify the solute and solvent in a solution?
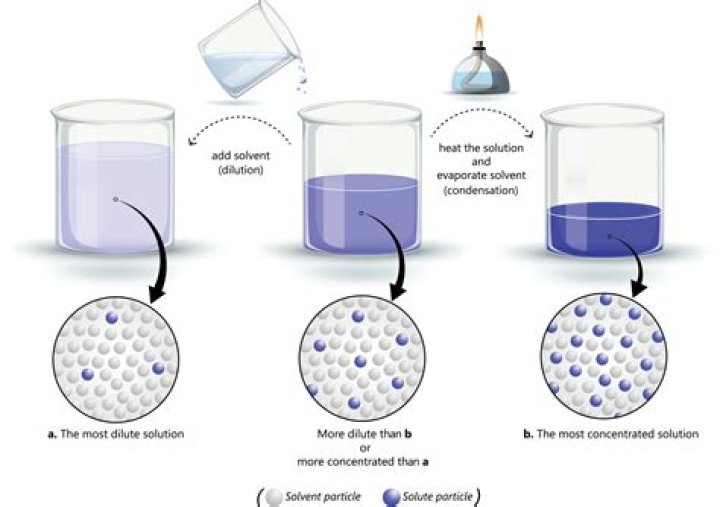
A solvent is a substance that dissolves the solute. And the solute is the substance being dissolved. To identify them, take note that if the subtance present in a solution undergoes change of state, it is the solute. However, if neither of them change state, the reagent in smaller amount is the solute.
Similarly, what is a mixture of solute and solvent called? A solution is a homogeneous mixture of a solute in a solvent. A solute is the material present in the smaller amount in the solution. A solvent is the material present in the larger amount in the solution.
One may also ask, what is the solvent solution?
A solvent is a liquid that dissolves a solid, liquid or gaseous solute. A solute is a substance dissolved in another substance. A solute and a solvent make up a solution. Water is the solvent and the salt is the solute and together they make a salt (saline) solution.
What is solute and solvent with example?
Some examples of solvents are water, ethanol, toluene, chloroform, acetone, milk, etc. Examples of solutes include, sugar, salt, oxygen, etc. There are numerous examples of solutions. For example milk (solvent) and sugar (solute) makes sweet milk.
Related Question AnswersWhat is solute and example?
noun. The definition of a solute is the substance you dissolve into a liquid. An example of a solute is sugar dissolving in hot water.What is the difference between a solute solvent and solution?
Difference Between Solute and Solvent. A solution can be defined as the homogenous mixture of two or more substances. So in a solution, the substance which gets dissolved is solute, whereas solvent is the substance in which the solute will dissolve.What are the solute and solvent in a solution?
A simple solution is basically two substances that are evenly mixed together. One of them is called the solute and the other is the solvent. A solute is the substance to be dissolved (sugar). The solvent is the one doing the dissolving (water).What is an example of a solution?
Some examples of solutions are salt water, rubbing alcohol, and sugar dissolved in water. In our example of salt water, the solute is the salt. Solvent: this is the substance that makes up the majority of the solution. This is the part where the solute is dissolved. In our example of salt water, the solvent is water.Is blood a solvent or solute?
Plasma (the liquid part of our blood) is made up of approximately 90% water. That's why the most common example of a solvent is water. Water is sometimes called the “universal solvent” because it is so good at dissolving a large variety of solutes.What is a hypotonic solution?
A hypotonic solution has a lower concentration of solutes than another solution. In biology, a solution outside of a cell is called hypotonic if it has a lower concentration of solutes relative to the cytosol. Due to osmotic pressure, water diffuses into the cell, and the cell often appears turgid, or bloated.Is water a solute or solution?
The solute is the substance which is dissolved by the solvent. For example, in a solution of salt and water, water is the solvent and salt is the solute. Solutions are formed because the molecules of the solute are attracted to the molecules of the solvent.How can you recognize a solution?
Characteristics- A solution is a homogeneous mixture of two or more substances.
- The particles of solute in a solution cannot be seen by the naked eye.
- A solution does not allow beams of light to scatter.
- A solution is stable.
- The solute from a solution cannot be separated by filtration (or mechanically).
What is the best solvent?
WaterWhat are the 10 example of solution?
Types of Solution| S.No | Types of Solution | Examples |
|---|---|---|
| 2 | Solid-liquid | The solution of sugar, salt etc in water. |
| 3 | Solid-gas | Sublimation of substances like iodine, camphor etc into the air. |
| 4 | Liquid-solid | Hydrated salts, mercury in amalgamated zinc etc. |
| 5 | Liquid-liquid | Alcohol in water, benzene in toluene |
What are the types of solvent?
There are 3 types of solvents commonly encountered: nonpolar, polar aprotic, and polar protic. (There ain't such a thing as a non-polar protic solvent).What is the role of a solvent?
Solvents play a vital role in occurrence of chemical reactions. It solvent's both the reagent and the reactant so that they can dissolve. In a chemical reaction, if the component are more polar than reactants then the polar solvent would accelerate the reaction. Some solvents are organic and some are inorganic.Whats is a solution?
A solution is a homogeneous mixture of two or more substances. A solution may exist in any phase. A solution consists of a solute and a solvent. The solute is the substance that is dissolved in the solvent. For example, in a saline solution, salt is the solute dissolved in water as the solvent.What is solvent made of?
A solvent is a substance that becomes a solution by dissolving a solid, liquid, or gaseous solute. A solvent is usually a liquid, but can also be a solid or gas. The most common solvent in everyday life is water. Most other commonly-used solvents are organic (carbon-containing) chemicals.What is the universal solvent?
A universal solvent is a substance that dissolves most chemicals. Water is called the universal solvent because it dissolves more substances than any other solvent. However, no solvent, including water, dissolves every chemical. Nonpolar solvents dissolve nonpolar molecules such as fats and other organic compounds.Is coffee a solute?
A solute is a substance that is dissolved in a solvent. Coffee is dissolved in hot water, (the solvent) and forms a solution (cup of coffee).What is the solute and solvent of Pepsi?
Coke or Pepsi has sugar, caffeine and carbon dioxide as solutes.What are the 9 types of solutions?
Terms in this set (9)- Solid Solute (Liquid) vinegar.
- Liquid Solute (Liquid) Salt water.
- Gas Solute (Liquid) Soft drink.
- Solid Solute (Gas) Mothballs.
- Liquid (Gas) humidity.
- Gas Solute (Gas) air.
- Solid Solute (Solid) gold-silver.
- Liquid Solute (Solid) dental filings.