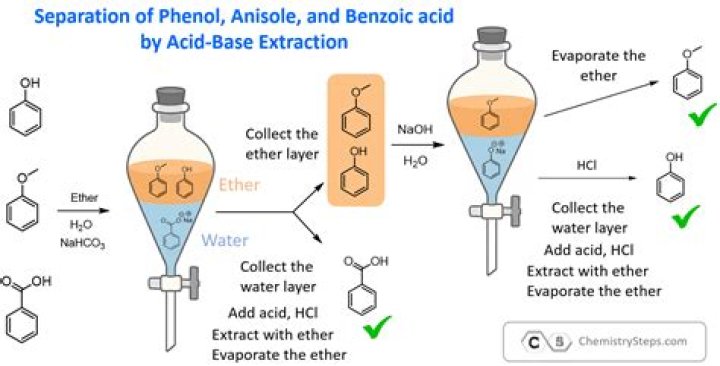
The idea behind an acid-base extraction is to utilize the acid-base properties of organic compounds and selectively isolate them from one another when they're present in a mixture. In organic chemistry, acids are known as carboxylic acids and contain the -COOH functional group..
Moreover, what is the purpose of an acid base extraction?
An acid-base extraction is a type of liquid-liquid extraction. Acid-base extraction is typically used to separate organic compounds from each other based on their acid-base properties. The method rests on the assumption that most organic compounds are more soluble in organic solvents than they are in water.
Also, what is the process of extraction? Extraction is a process in which one or more components are separated selectively from a liquid or solid mixture, the feed (Phase 1), by means of a liquid immiscible solvent (Phase 2). Afterwards in order to regenerate the solvent, another separation step (e.g. distillation) is finally required.
Also Know, why is Naoh used in extraction?
This phenomenon will often be observed if sodium bicarbonate is used for the extraction in order to neutralize or remove acidic compounds. The reaction affords carbon dioxide (CO2), which is a gas at ambient temperature. Pressure builds up that pushes some of the gas and the liquid out.
How do you tell an acid from a base?
To determine whether a substance is an acid or a base, count the hydrogens on each substance before and after the reaction. If the number of hydrogens has decreased that substance is the acid (donates hydrogen ions). If the number of hydrogens has increased that substance is the base (accepts hydrogen ions).
Related Question Answers
What is acid base extraction used for?
Acid-base extraction is typically used to separate organic compounds from each other based on their acid-base properties.What is extraction used for in real life?
The process of steeping leaves or other plant parts in hot water is an example of extraction. Anything in the plants that is soluble in hot water will pass into your beverage, while anything that is not soluble in hot water (cellulose, chlorophyll, other hydrophobic molecules) stays with the solid matter.Is NaOH an acid or base?
NaOH is a base because when dissolved in water it dissociates into Na+ and OH- ions. It is the OH- (hydroxyl ion) which makes NaOH a base. In classical term a base is defined as a compound which reacts with an acid to form salt and water as depicted by the following equation. NaOH+HCl=NaCl+H2O.Why is nahco3 a weak base?
Sodium bicarbonate is a weak base which is commonly known as baking soda and used in cooking. It weakly ionizes in water: In reaction with an acid, it liberates carbon dioxide gas: This reaction is an important part of the leavening which takes place in baking.How do you extract carboxylic acids?
As previously discussed, carboxylic acids can be extracted from an organic layer into an aqueous layer by shaking them with basic solutions, which converts them into their more water-soluble salts. A similar reaction occurs with phenols (PhOH), and they too can be extracted into an aqueous NaOH layer (Figure 4.58a).Is NaOH a strong base?
Sodium hydroxide (NaOH) is strong base because it fully dissociates in water to produce hydroxide ions. While ammonia (NH3) is weak base because it accepts protons from water to produce fewer hydroxide ions in solution. While weak bases produce fewer hydroxide ions, making the solution less basic.What makes a strong acid?
A strong acid is any acid that ionizes completely in solutions. This means it gives off the greatest number of hydrogen ions or protons when placed in a solution. Ions are charged particles. This means that it has one proton, and therefore a hydrogen ion is essentially a proton.Why is HCl a strong acid?
Dissociation of acids When HCl molecules dissolve they dissociate into H+ ions and Cl- ions. HCl is a strong acid because it dissociates almost completely. By contrast, a weak acid like acetic acid (CH3COOH) does not dissociate well in water – many H+ ions remain bound-up within the molecule.Why is extraction important?
There are several reasons to use extraction in the chemistry lab. It is a principal method for isolating compounds from plant materials. Extraction moves compounds from one liquid to another, so that they can be more easily manipulated or concentrated. It also enables the selective removal of components in a mixture.What is extraction theory?
Extraction Theory. Back. Theory of Liquid-Liquid Extraction. As we saw in the previous lab, in chemistry, extraction is the physical process by which a compound (or a mixture of compounds) is transferred from one phase into another. The isolation of trimyristin form nutmeg is an example of a solid-liquid extraction.Why is multiple extraction more efficient?
So with multiple extractions the amount of material left in the residue will be lower, ergo the extraction will be more complete. Several extractions with smaller volumes of solvent are more effective than a single extraction with a large volume of solvent.Why is DCM used for extraction?
The DCM is the desired solvent to be harvested in our extraction because it has the majority of caffeine dissolved in it. The DCM extract is collected from the funnel and more DCM is added and the process is repeated as a second extraction. This increases our overall yield.Why is brine used in extraction?
The bulk of the water can often be removed by shaking or "washing" the organic layer with saturated aqueous sodium chloride (otherwise known as brine). This is because the concentrated salt solution wants to become more dilute and because salts have a stronger attraction to water than to organic solvents.Why do emulsions form during extraction?
Emulsions often occur with samples where the animal (or human) diet is high in fats. By swirling the separatory funnel the agitation that can cause the emulsion to form is reduced, but the surface area of contact between the two phases is maintained to allow for extraction to occur.Is NaOH polar or nonpolar?
Usually nonpolar covalent, polar covalent, and ionic. As sodium hydroxide has a metal and nonmetallic mix, we can assume that between the sodium and oxygen there is an ionic interaction. And between the hydrogen and oxygen, it's extreme but not as extreme. This is just very polar.What happens when you add NaOH to benzoic acid?
NaOH is a base. The H from the OH of the benzoic acid combines with the OH of NaOH to form water (H2O). The Na+ cation joins the O- of the benzoic acid. Benzoic acid (C7H6O2 or C6H5-COO-H) react with Na-OH and produce a salt, the Sodium benzoate (and water).Why is NaOH heated before acidification?
(c) Why is the NaOH extract heated before acidification? The NaOH extract is heated before acidification to remove any traces of tert-butyl methyl ether which might inhibit the crystallization of the phenol.What is an example of extraction?
Extraction is the process of selectively removing a compound of interest from a mixture using a solvent. Making tea is a good example of extraction. Water is placed in contact with tea bags and the "tea" is extracted from the tea leaves into the water.What is the synonym of extraction?
Choose the Right Synonym for extract Verb. educe, evoke, elicit, extract, extort mean to draw out something hidden, latent, or reserved. educe implies the bringing out of something potential or latent.