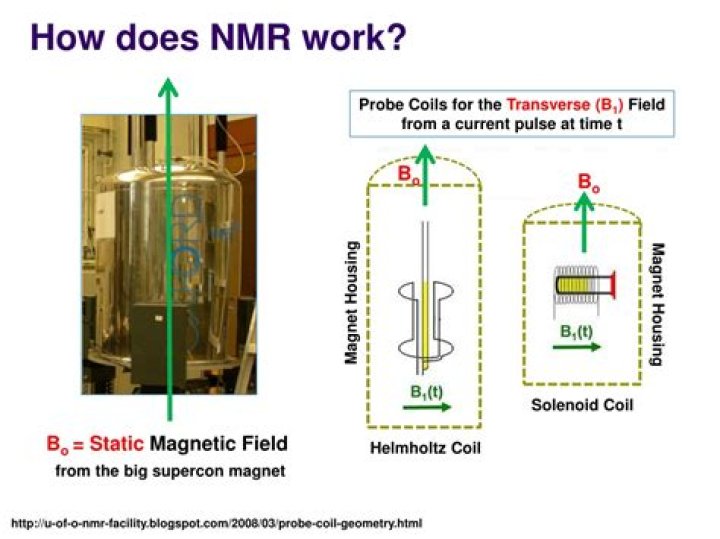
The NMR Spectrometer A stable magnet with a sensitive controller to produce a precise magnetic field. A radio-frequency (RF) transmitter, emitting a precise frequency (continuous wave or CW) A detector to measure the same's absorption of RF energy..
Regarding this, how does an NMR work?
The principle behind NMR is that many nuclei have spin and all nuclei are electrically charged. The energy transfer takes place at a wavelength that corresponds to radio frequencies and when the spin returns to its base level, energy is emitted at the same frequency.
Subsequently, question is, is NMR emission or absorption spectroscopy? IR and NMR spectroscopy are two forms of absorption spectroscopy. Emission spectroscopy. This type of spectroscopy uses an electromagnetic spectrum that a substance emits rather than absorbs, such as luminescence.
Keeping this in view, how is an NMR signal generated?
The sample is placed in a magnetic field and the NMR signal is produced by excitation of the nuclei sample with radio waves into nuclear magnetic resonance, which is detected with sensitive radio receivers. NMR spectra are unique, well-resolved, analytically tractable and often highly predictable for small molecules.
How much does an NMR cost?
A standard 600 MHz NMR costs roughly $800,000, but the 900 MHz sells for about $5 million.
Related Question Answers
How is NMR measured?
NMR is an abbreviation for Nuclear Magnetic Resonance. An NMR instrument allows the molecular structure of a material to be analyzed by observing and measuring the interaction of nuclear spins when placed in a powerful magnetic field.Where is NMR used?
Nuclear magnetic resonance spectroscopy is widely used to determine the structure of organic molecules in solution and study molecular physics, crystals as well as non-crystalline materials. NMR is also routinely used in advanced medical imaging techniques, such as in magnetic resonance imaging (MRI).Why is TMS used in NMR?
Uses in NMR spectroscopy Because of its high volatility, TMS can easily be evaporated, which is convenient for recovery of samples analyzed by NMR spectroscopy. Because all twelve hydrogen atoms in a tetramethylsilane molecule are equivalent, its 1H NMR spectrum consists of a singlet.How many NMR signals are there?
three signals
What solvent is used in NMR?
NMR solvents are distinctly different from other spectroscopic solvents as majority of hydrogen nuclei are replaced with deuterium so as to minimize the interference due to protons. The price increases with the degree of deuteration. Deuterated chloroform, is most commonly used because of its low price.Does a nucleus spin?
The nucleus has a positive charge and is spinning. This generates a small magnetic field. The nucleus therefore possesses a magnetic moment, m, which is proportional to its spin,I. The constant, g, is called the magnetogyric ratioand is a fundamental nuclear constant which has a different value for every nucleus.Who invented NMR?
Isidor Rabi
What is chemical shift in NMR?
From Wikipedia, the free encyclopedia. In nuclear magnetic resonance (NMR) spectroscopy, the chemical shift is the resonant frequency of a nucleus relative to a standard in a magnetic field. Often the position and number of chemical shifts are diagnostic of the structure of a molecule.What does 1h NMR tell?
Proton nuclear magnetic resonance (proton NMR, hydrogen-1 NMR, or 1H NMR) is the application of nuclear magnetic resonance in NMR spectroscopy with respect to hydrogen-1 nuclei within the molecules of a substance, in order to determine the structure of its molecules.What type of nuclei are NMR active?
NMR occurs due to the absorbance of radio frequency radiation to cause the "flipping" of nuclear spins from low to high energy spin states. While not all nuclei are NMR active (e.g. 12C and 16O are inactive), the most important nuclei for organic chemists are 1H and 13C (both with nuclear spin = 1/2).What is NMR in construction?
NMR or Nominal Muster Roll means any `daily wage employee' or any person who is employed on the basis of payment of daily wages.What is spin flipping in NMR?
In the presence of the static magnetic field which produces a small amount of spin polarization, a radio frequency signal of the proper frequency can induce a transition between spin states. This "spin flip" places some of the spins in their higher energy state. This process is called Nuclear Magnetic Resonance (NMR).What are signals in NMR?
? NMR signal: Peaks in the NMR spectrum plot. ? Proton: Particle or atom bearing a positive (+) charge, H is referred to as a proton. ? Equivalent protons: Protons with magnetic environment that are identical in EVERY. way, can't be distinguished from each other based on relative position.What is NMR testing?
The NMR LipoProfile test is an advanced cardiovascular diagnostic test that uses nuclear magnetic resonance (NMR) spectroscopy to provide rapid, simultaneous and direct measurement of LDL particle number and size of LDL particles, and also direct measurement of HDL and VLDL subclasses.What is coupling in NMR?
It is an indirect interaction between two nuclear spins which arises from hyperfine interactions between the nuclei and local electrons. In NMR spectroscopy J-coupling contains information about relative bond distances and angles. Most importantly, J-coupling provides information on the connectivity of chemical bonds.What does NMR mean?
Not My Responsibility
What is meant by coupling constant?
Coupling Constant. The coupling constant is defined as nJA,X, where n is the number of chemical bonds between the two coupling atoms A and X. The coupling constant is independent of the field strength, and has a plus or minus prefix and it is mutual to the coupled atoms (nJA,X=nJX,A).What is the type of electromagnetic radiation used in NMR?
Like all spectroscopies, NMR uses a component of electromagnetic radiation (radio frequency waves) to promote transitions between nuclear energy levels (Resonance). Most chemists use NMR for structure determination of small molecules.What is Spin spin splitting?
identification of organic compounds atoms through a process termed spin-spin splitting. Each set of equivalent hydrogens on a given carbon is split into an n+1 multiplet by adjacent hydrogen atoms that are nonequivalent to the hydrogens of the given carbon.