.
Similarly, how is sublimation helpful in daily life?
There are many examples of sublimation in daily life: The air fresheners used in toilets. The solid slowly sublimes and releases the pleasant smell in the toilet over a certain period of time. Dry ice, or solid carbon dioxide is used for cooling items, store special adhesives and also used for neutralizing alkalis.
what are the advantages of sublimation? The advantage of digital sublimation printing is that no plates are required, so it is ideal for customizing designs with changing elements such as names, numbers, etc. Sublimation is quick and easy to produce, allowing for same day or same hour printing, as well as on demand printing that reduces risk and wastage.
Hereof, why is sublimation important?
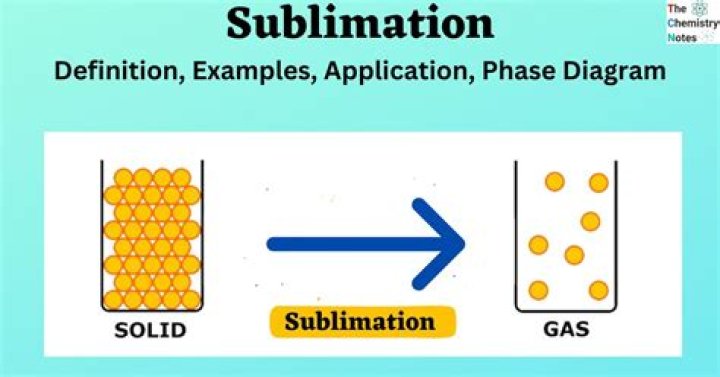
Sublimation is the phase transition of a substance directly from the solid to the gas phase without passing through the intermediate liquid phase. This technology of sublimation can help people highly improve the efficiency of working and save a lot of time and money.
What are the application of sublimation?
Used in purifying processes,used in making naphthalene balls,used in room freshners and in drying processes without leaving any residue. items include mugs, phone cases, keychains, photo slates and so on. Fabrics material and polyester such as t-shirt and etc.
Related Question AnswersWhat are 3 examples of sublimation?
Sublimation Examples in Real Life- Dry Ice. As mentioned earlier, dry ice is one of the most popular examples of sublimation in real life.
- Water.
- Specialized Printers.
- Moth Balls.
- Freeze Drying.
- Air Fresheners.
What is a good example of sublimation?
Through sublimation, a substance changes from a solid to a gas without ever passing through a liquid phase. Dry ice, solid CO2, provides a common example of sublimation. It is also possible for ice to sublimate, though it requires specific weather environments and high altitudes.What is the basic principle of sublimation?
Sublimation is a process in which a solid turns directly into gas without passing into liquid state. It works on the principle that solids have a weak intermolecular force hence a higher vapour pressure which converts it into directly vapour state.Is snow an example of sublimation?
Sublimation is the process in which a solid changes directly to a gas without going through the liquid state. Solid carbon dioxide is an example of a substance that undergoes sublimation. Snow and ice undergo sublimation under certain conditions.What materials can sublimate?
Typically solids undergo sublimation at low pressures (under vacuum). At standard, atmospheric pressure, a few solids which will sublime are iodine (at slightly higher than room temperature), carbon dioxide (dry ice) at -78.5 degrees Celsius, as well as naphthalene (used in mothballs) and arsenic.Is Iodine an example of sublimation?
Iodine provides an excellent example of sublimation. When iodine sublimes it forms a visible purple gas. When the gas cools it returns directly to the solid phase, an example of deposition.Where does sublimation occur on Earth?
Public domain. Sublimation occurs more readily when certain weather conditions are present, such as low relative humidity and dry winds. Sublimation also occurs more at higher altitudes, where the air pressure is less than at lower altitudes. Energy, such as strong sunlight, is also needed.Does sublimation occur in nature?
When sublimation occurs, the substance does not go through the liquid phase. Energy is required for a solid to sublime into a gas. In nature, the heat produced by sunlight is usually the energy source. An example of sublimation is how dry ice reacts when exposed to an average room temperature and pressure.What is the opposite of sublimation?
Sublimation is the transition from the solid to the gas state. A good example is carbon dioxide ice. The opposite of this process is called deposition : the transition from the gas to the solid. Condensation is the transition from gas to liquid as in the condensation of steam to liquid water.What happens during sublimation?
When energy is transferred to dry ice, the solid carbon dioxide does not melt to liquid carbon dioxide. Instead, the solid changes directly to a gas. This process is called sublimation. Sublimation occurs when molecules of a solid move fast enough to overcome the attractions from other molecules and become a gas.How do you spell sublimation?
Correct spelling for the English word "sublimation" is [s_ˌ?_b_l_?_m_ˈe?_?_?_n], [sˌ?bl?mˈe????n], [sˌ?bl?mˈe???n]] (IPA phonetic alphabet).Similar spelling words for SUBLIMATION
- Sublime Porte,
- sublimate,
- sublimated,
- sublimed,
- sublimity,
- Sublimaze,
- suppliant,
- subliminal,
What would happen if sublimation didn't occur?
It would change directly to a gas without going through the liquid state. The process in which a solid changes directly to a gas is called sublimation. It occurs when energy is added to a solid such as dry ice.How will you show sublimation process What is the importance of this process?
Sublimation: Sublimation is the process of separation of a mixture of two or more solids in which one of the solid should be sublime easily. Importance of Sublimation Process: Like ammonium chloride is a sublime substance so it can easily separate from sand or salt which are not sublime substances.Does sublimation release energy?
Energy is required to change from solid to liquid, liquid to gas (evaporation), or solid to gas (sublimation). Energy will be released to change from liquid to solid (fusion), gas to liquid (condensation), or gas to solid.Does sublimation require heat?
The enthalpy of sublimation, or heat of sublimation, is the heat required to change one mole of a substance from solid state to gaseous state at a given combination of temperature and pressure, usually standard temperature and pressure (STP).What are the disadvantages of sublimation?
Disadvantages of sublimation 1. Recovery may not be complete - the fumes may be blown away. 2. Non-sublimable agent may decompose under heat.What are the limitations for sublimation?
Some of the limitations of the dye-sublimation transfer process are:- Profiles are specific to the fabric. End users must have access to a dealer that is able to set them up with a specific profile to their substrate.
- Investment cost is not only determined by the printer and supplies.