.
Subsequently, one may also ask, how many subunits does immunoglobulin have?
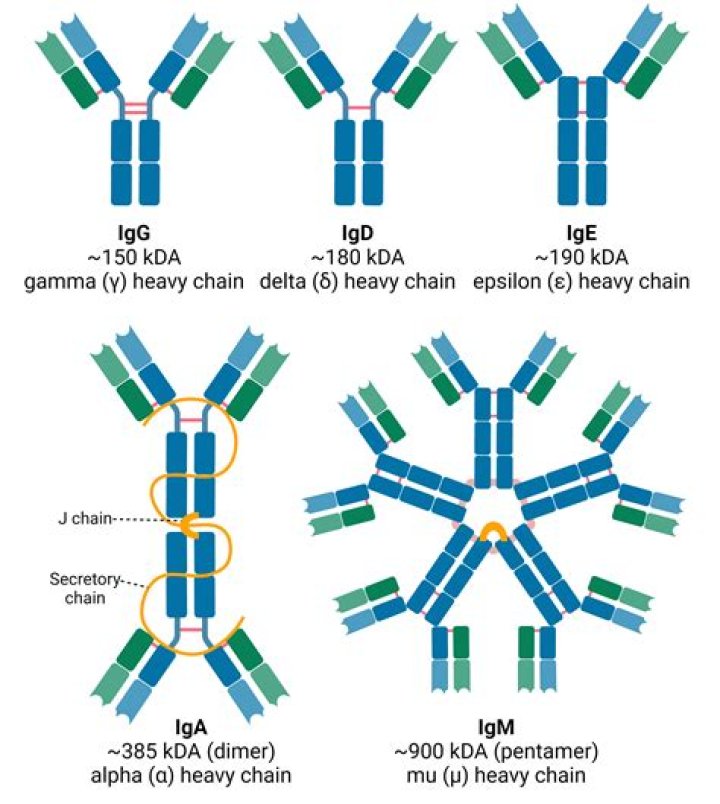
The major form in serum is the 19S, 900 000 Da pentameric IgM, which contains five subunits [(µ2L2)5] linked together by disulfide bridges and by one molecule of an additional polypeptide chain, the J chain, which joins two of the subunits by a disulfide bridge.
Secondly, what is an Ig fold? Definition. Immunoglobulin-fold designates a protein domain structure, first discovered in immunoglobulin constant and variable domains, which consists of two β-sheets packed against each other.
Then, how many Ig domains does an antibody have?
Several immunoglobulin domains make up the two heavy chains (red and blue) and the two light chains (green and yellow) of an antibody. The immunoglobulin domains are composed of between 7 (for constant domains) and 9 (for variable domains) β-strands.
What are designated as immunoglobulin domain and immunoglobulin fold?
An immunoglobulin domain consists of a pair of β-sheets linked by a disulfide bond and hydrophobic interactions. The immunoglobulin fold consists of a pair of β sheets, each built of antiparallel β strands, that surround a central hydrophobic core. A single disulfide bond bridges the two sheets.
Related Question AnswersWhat are the 5 types of immunoglobulins and what are their functions?
Often abbreviated as "Ig," antibodies are found in blood and other bodily fluids of humans and other vertebrate animals. They help identify and destroy foreign substances such as microbes (e.g., bacteria, protozoan parasites and viruses). Immunoglobulins are classified into five categories: IgA, IgD, IgE, IgG and IgM.What is heavy and light chain?
IgG antibodies are large molecules, having a molecular weight of approximately 150 kDa, composed of two different kinds of polypeptide chain. One, of approximately 50 kDa, is termed the heavy or H chain, and the other, of 25 kDa, is termed the light or L chain (Fig. 3.2).What are the 5 immunoglobulins?
There are five immunoglobulin classes (isotypes) of antibody molecules found in serum: IgG, IgM, IgA, IgE and IgD. They are distinguished by the type of heavy chain they contain. IgG molecules possess heavy chains known as γ-chains; IgMs have μ-chains; IgAs have α-chains; IgEs have ε-chains; and IgDs have δ-chains.What is the basic structure of an antibody?
Basic structure of an antibody. The basic structure of an antibody molecule contains four polypeptide chains, two identical light chains or L chains, which are each made up of ca. 220 amino acids (AA), and two identical heavy chains or H chains that are built up from ca.What is the structure of IgG?
Structure. IgG antibodies are large globular proteins with a molecular weight of about 150 kDa made of four peptide chains. It contains two identical γ (gamma) heavy chains of about 50 kDa and two identical light chains of about 25 kDa, thus a tetrameric quaternary structure.Is IgG a protein?
Immunoglobulin G. Immunoglobulin G (IgG) is a heteromeric protein composed of two different gene products that are known as heavy and light chains.What are the functions of immunoglobulins?
Immunoglobulins, also known as antibodies, are glycoprotein molecules produced by plasma cells (white blood cells). They act as a critical part of the immune response by specifically recognizing and binding to particular antigens, such as bacteria or viruses, and aiding in their destruction.What type of protein is immunoglobulin?
Immunoglobulins Proteins. Immunoglobulin, also known as antibody, is large, Y-shape glycoprotein molecules produced by plasma cells in identification and response to an immunogen such as bacteria and viruses. Immunoglobulin or antibodies play vital roles in immune defenses.What are the four functions of antibodies?
Major functions of the antibodies are:- Neutralization of infectivity,
- Phagocytosis,
- Antibody-dependent cellular cytotoxicity (ADCC),
- Complement-mediated lysis of pathogens or of infected cells: Antibodies activate the complement system to destroy bacterial cells by lysis.