Solubility : Soluble in water (13.5 mg/ml at 25° C), methanol (almost transparency), pyridine (303.03 mg/ml), ethanol (166.67 mg/ml), and ethyl ether (slightly)..
Moreover, what is benzamide soluble in?
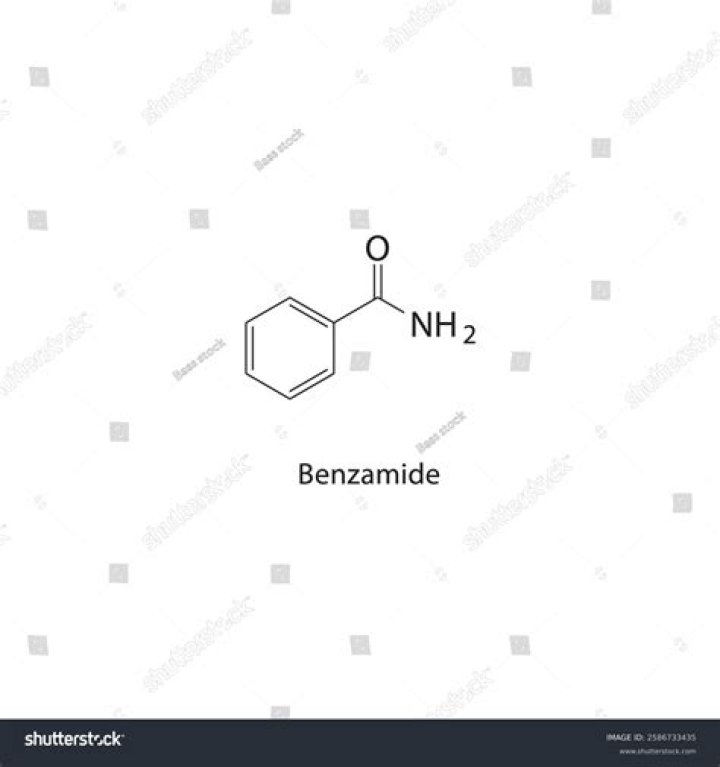
Benzamide is a white solid with the chemical formula of C6H5C(O)NH2. It is the simplest amide derivative of benzoic acid. It is slightly soluble in water, and soluble in many organic solvents. A number of substituted benzamides exist.
Additionally, is benzamide flammable? SYMPTOMS: This compound may produce gastric pain, nausea, and vomiting. BENZAMIDE reacts with azo and diazo compounds to generate toxic gases. Forms flammable gases with strong reducing agents. Combustion generates toxic mixed oxides of nitrogen (NOx).
In this way, is benzamide soluble in diethyl ether?
Aniline, benzoic acid and benzamide are all insoluble in water, but soluble in ether. Explain how, by using simple laboratory reagents and equipment, each compound could be separated and recovered from a mixture of all three. All of the compounds are neutral and will dissolve in an organic solvent like ether.
What is the formula of benzamide?
C7H7NO
Related Question Answers
Is benzamide water soluble?
Benzamide is a white solid with the chemical formula of C6H5C(O)NH2. It is the simplest amide derivative of benzoic acid. It is slightly soluble in water, and soluble in many organic solvents.What is benzamide used for?
Benzamides are a class of chemical compounds derived from Benzamid, the carbonic acid amide of benzoic acid. In psychiatry some substituted benzamides are therapeutically used as neuroleptics and/or antipsychotics (wikipedia).What happens when benzamide is heated?
When benzamide is heated with dilute mineral acid, benzoic acid is obtained. When benzamide is heated with dilute mineral acid we get benzoic acid and a salt ammonium chloride as products. The acid act as catalyst for reaction between amide and water thus increasing rate of reaction.What is the melting point of benzamide?
Chemical Properties Benzamide is a combustible, colorless to beige, off-white, crystalline solid; freezing/melting point=132 133° C.Is benzamide a base?
Benzamide, also known as benzoate amide or PHC(=o)NH2, belongs to the class of organic compounds known as benzamides. Benzamide is an extremely weak basic (essentially neutral) compound (based on its pKa). Benzamide exists in all living organisms, ranging from bacteria to humans. Benzamide is a bitter.Is benzoic acid soluble in water?
Benzoic Acid. Benzoic acid or benzene-carbonic-acid is a monobasic aromatic acid, moderately strong, white crystalline powder, very soluble in alcohol, ether, and benzene, but poorly soluble in water (0.3 g of benzoic acid in 100 g of water at 20 °C).Why is benzoic acid soluble in sodium bicarbonate?
small organic molecules are more soluble in water than are large organic molecules; 2. For example, benzoic acid is not soluble in water, yet it is soluble in sodium hydroxide solution and in sodium hydrogen carbonate solution because these bases react with benzoic acid to form the water-soluble benzoate ion.How do you convert benzamide to Benzylamine?
Benzamide can be converted into benzylamine using $$LiAl{H}_{4}$$. The more reactive $$LiAl{H}_{4}$$ is required for the reduction of the amide to the corresponding amine.Is benzaldehyde soluble in h2so4?
2) aromatic carbonyl compounds like benzaldehyde and acetophenone are NOT soluble in H2O, 5%NaOH, 5% NaHCO3, 5% HCl. But they are soluble in highly concentrated H2SO4.Is Benzil soluble in NaOH?
Benzil is soluble in an organic solvent, such as diethyl ether or ethanol, but insoluble in polar solvents, like water and NaOH. No, Benzil is NOT soluble in NaOH.What is solubility classification?
Solubility. Solubility is a chemical property referring to the ability for a given substance, the solute, to dissolve in a solvent. It is measured in terms of the maximum amount of solute dissolved in a solvent at equilibrium. The resulting solution is called a saturated solution.Is benzophenone soluble in HCl?
Benzophenone, which is largely nonpolar, but possesses a polar carbonyl group, was found to be partially soluble in methyl alcohol and hexane but insoluble in water. Benzoic acid was found to be soluble in water and 1.0 M NaOH, however upon addition of 6.0 M HCl to this solution, benzoic acid became insoluble.Why is benzoic acid soluble in NaOH?
Benzoic acid is a weak acid, thus it will only partially dissolve in water. While NaOH is a strong base, it will attack the weak acid and form a salt. Benzoic acid is a weak acid, thus it will only partially dissolve in water. While NaOH is a strong base, it will attack the weak acid and form a salt.What is the main precaution when handling benzamide?
7.1 Precautions for safe handling Avoid contact with skin and eyes. Avoid formation of dust and aerosols. Provide appropriate exhaust ventilation at places where dust is formed.Is ethanol soluble in NaHCO3?
2 Answers. Firstly ethanol being an organic compound, tends to dissolve less in polar compounds. But it can form numerous hydrogen bonds in water and hence is soluble in most aqueous solutions. That is not the case with NaHCO3 as its an even weaker base, so it rarely participates in any reaction with ethanol.What compounds are soluble in NaOH?
Water Ethanol MethanolIs oxalic acid soluble in NaOH?
Is oxalic acid soluble in NaOH? “ soluble “ , no. It will react with it so you can't make the statement that it just dissolves in it. It dissolves in water for 9 to 10% at 20°C.How do you convert benzamide to benzoic acid?
Benzamide is hydrolyzed to sodium benzoate by boiling with a sodium hydroxide solution. Sodium benzoate on acidification gives benzoic acid. To prepare benzoic acid start with 3 grams of benzamide and mix it with 2 grams of sodium hydroxide dissolved in about 18 mL of water in a round bottom flask..What organic compounds can be oxidized to produce benzoic acid?
Benzyl alcohol and benzyl chloride and virtually all benzyl derivatives are readily oxidized to benzoic acid.