.
People also ask, is hno3 a strong or weak acid?
The 7 common strong acids are: HCl, HBr, HI, HNO3, HClO3, HClO4 and H2SO4 (1st proton only). Remember what we mean by a weak acid or weak base. They simply do not ionize completely whereas a strong acid or base does.
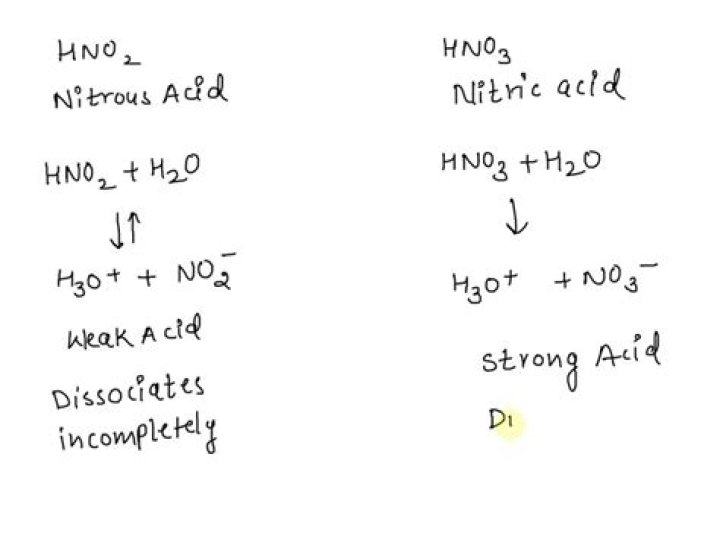
is hno2 an acid? No it is not a strong acid. HNO2 is a weak acid.
In this way, why is hno2 a weak acid and hno3 a strong acid?
If you were familiar with the list of 7 strong acids, then you'd know that HNO3 is a strong acid, and HNO2 is a weak acid. Thus, HNO3 would be stronger. When an acid dissociates, the stability of the products can tell you how likely the dissociation can happen. For example, HA -> H+ + A-.
Is h2co3 a strong or weak acid?
- Quora. conjugate base of HCl is a weak base Cl-. thus when we examine the carbonic acid, H2CO3, when it loses a proton, H+, the base formed will be a strong and resonance staballized base CO3- -. Thus it is aweak acid.
Related Question AnswersWhat are the 7 strong acids?
There are 7 strong acids: chloric acid, hydrobromic acid, hydrochloric acid, hydroiodic acid, nitric acid, perchloric acid, and sulfuric acid. Being part of the list of strong acids doesn't give any indication of how dangerous or damaging an acid is though.What are the six strong acids?
They are H2SO4 (or sulfuric acid), HI (hydrologic acid), HBr (hydrobromic acid), HNO3 (nitric acid), HCl (hydrochloric acid) and HClO4 (perchloric acid). The mnemonic that I can use to help you memorize these six strong acids is: So I Brought No Clean Clothes.Is HCl strong or weak?
Because HCl is a strong acid, its conjugate base (Cl−) is extremely weak. The chloride ion is incapable of accepting the H+ ion and becoming HCl again. In general, the stronger the acid, the weaker its conjugate base.Is LiOH strong or weak?
Classifying Electrolytes| Strong Electrolytes | strong acids | HCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4 |
|---|---|---|
| strong bases | NaOH, KOH, LiOH, Ba(OH)2, and Ca(OH)2 | |
| salts | NaCl, KBr, MgCl2, and many, many more | |
| Weak Electrolytes | ||
| weak acids | HF, HC2H3O2 (acetic acid), H2CO3 (carbonic acid), H3PO4 (phosphoric acid), and many more |
Is koh a strong acid?
KOH is potassium hydroxide. Since it is composed of the hydroxide anion (OH-), it is a strong base. In solution, the hydroxide anion will completely react with any available protons, that is why KOH is a strong base. It is not an acid of any type, weak or strong, since KOH does not contribute any protons to solution.What are the strong bases?
Strong bases are able to completely dissociate in water- LiOH - lithium hydroxide.
- NaOH - sodium hydroxide.
- KOH - potassium hydroxide.
- RbOH - rubidium hydroxide.
- CsOH - cesium hydroxide.
- *Ca(OH)2 - calcium hydroxide.
- *Sr(OH)2 - strontium hydroxide.
- *Ba(OH)2 - barium hydroxide.
Is h3po4 a strong or weak acid?
Along with the higher electronegativity of N relative to P, the extra oxygen makes HNO3 a strong acid, while the lack thereof makes H3PO4 weak. After H3PO4 does lose a proton, it forms H2PO4-, which lacks sufficient resonance stabilization. The more resonance structures a molecule has, the more stable it is.Is HF stronger than HCl?
HCl is a stronger acid as compared to HF. A molecule that produces more protons in a solution is more acidic. If we consider the electronegativity differences, the H-F bond should be weaker and thus cleave easily to give more of H+ in a solution, making HF a stronger acid.Is NaCl an acid or base?
Expert Answers info NaCl is formed by the reaction of HCl and NaOH. Both are strong acids and bases. When a strong acid and a strong base react together the resultant is salt and water. Therefore NaCl is a salt.Is NaOH an acid or base?
NaOH is a base because when dissolved in water it dissociates into Na+ and OH- ions. It is the OH- (hydroxyl ion) which makes NaOH a base. In classical term a base is defined as a compound which reacts with an acid to form salt and water as depicted by the following equation.Is no2 an acid or base?
NO2 IS AN ACIDIC OXIDE. IT IS BASICALLY AN ACID ANHYDRIDE OF BOTH HNO3(NITRIC ACID) AND HNO2(NITROUS ACID).Is ch3cooh an acid or base?
CH3COOH is a weak acid and dissociates partially in solution (as indicated with reversible arrow) to form H+ and CH3COO- ions. Since this is a reversible process, CH3COO- can accept H+ to form back CH3COOH. Therefore the nature of CH3COO- is basic and we call CH3COO- the conjugate base of CH3COOH.Is HCl a strong electrolyte?
Hydrochloric acid, HCl(aq), dissociates completely so there are many ions in solution to conduct electricity therefore hydrochloric acid is a strong electrolyte. State your solution to the problem "is hydrochloric acid a strong or weak electrolyte": Hydrochloric acid is a strong electrolyte.Is h2po4 strong or weak?
More on Acid and Base| A | B |
|---|---|
| H2CO3, H2PO4-, NH4+, HCN, HCO3-, HPO42- | Very weak acids |
| HCO3-, HPO42-, NH3, CN, CO32-, PO43- | Weak bases |
| Strong Acids | Give up proton easily and are essentially 100% dissociated in water |
| Weak Acids | give up proton with difficulty and are less than 100% dissociated in water |