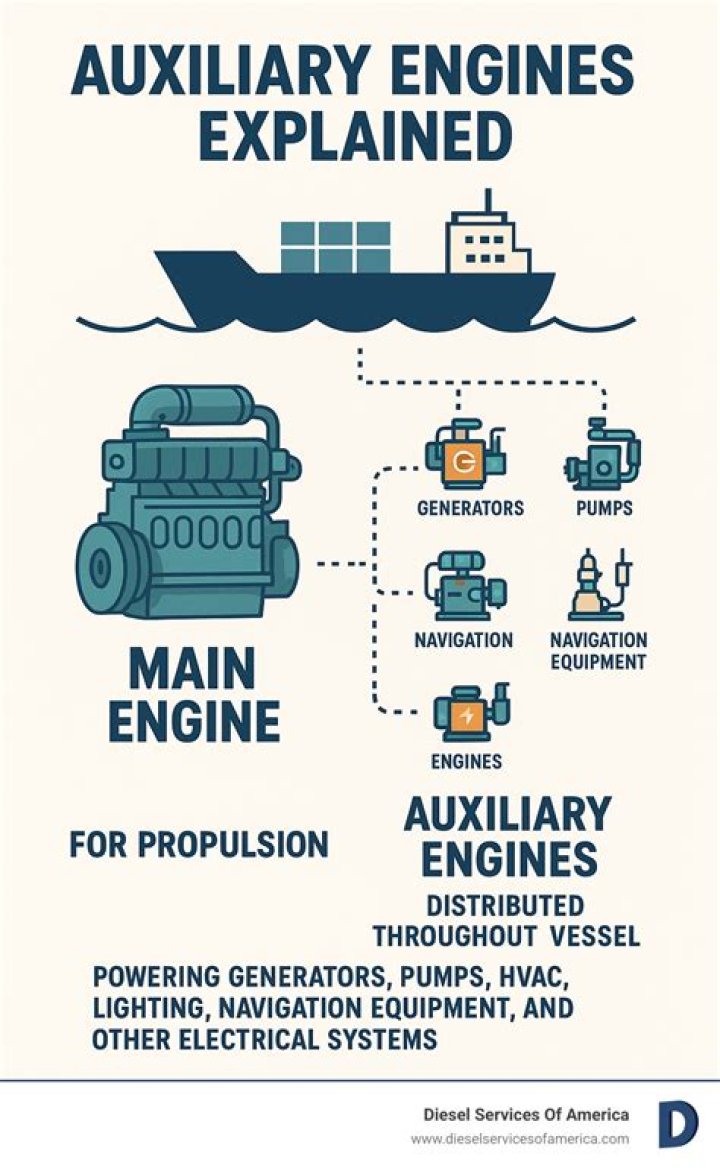
Some nitrogen dioxide is formed naturally in the atmosphere by lightning and some is produced by plants, soil and water. The major source of nitrogen dioxide in Australia is the burning of fossil fuels: coal, oil and gas. Most of the nitrogen dioxide in cities comes from motor vehicle exhaust (about 80%)..
Similarly, is no2 gas soluble in water?
Water: Nitrogen oxides are only slightly soluble in water but they form nitrous or nitric acid when they come into contact with water. In Germany, nitrogen dioxide is listed in water hazard class 1.
Additionally, is nitrogen soluble in water? Nitrogen gas does not react with water. It does dissolve in water. Nitrogen (N2) solubility at 20oC and pressure = 1 bar is approximately 20 mg/L. Nitrates and ammonia dissolve in water readily.
Also Know, what happens when nitrogen dioxide dissolves in water?
When nitrogen dioxide reacts with water, Mono-nitrogen oxides eventually form nitric acid when dissolved in atmospheric moisture, forming a component of acid rain.
Is nitrogen dioxide a mixture?
Oxides of nitrogen are a mixture of gases that are composed of nitrogen and oxygen. Two of the most toxicologically significant compounds are nitric oxide (NO) and nitrogen dioxide (NO2). Other gases belonging to this group are nitrogen monoxide (or nitrous oxide, N2O), and nitrogen pentoxide (NO5).
Related Question Answers
Is c6h14 soluble in water?
N-hexane is a clear colorless liquids with a petroleum-like odor. Flash points -9°F. Less dense than water and insoluble in water.Is oxygen soluble in water?
1 Answer. Oxygen is relatively insoluble in water, its solubility being only 264 μM at 25oC.Is no2 polar or nonpolar?
NO2 is a polar molecule. The geometry of the molecule is bent because of a non-bonding pair of electrons. The bent geometry causes the polarity. Polarity arises from a difference in electronegativity. Oxygen is more electronegative than nitrogen and pulls the electrons from the bonds closer.Is hydrogen soluble in water?
Hydrogen gas is weakly soluble in water. Hydogen gas does not have a permanent dipole moment and does not dissolve well in water which is highly polar. But H2 has a relatively high polarizability, which makes it weakly soluble in water.Why is no2 Brown in Colour?
Why is nitrogen oxide colourless while nitrogen dioxide is brown in colour? That is why we see that Nitrogen gas is a white gas(if the gas if concentrated enough). As for nitrogen dioxide, the molecules absorb a certain color. All the other colors are reflected as a brown color.Is no2 linear?
Just like carbon dioxide? Except that NO2 is a V-shaped molecule, and CO2 is linear. two N=O double bonds and no unpaired electrons, so repulsion between the two regions of electron density is minimised by the 180° bond angle, and it is linear, as with CO2.Is no2 poisonous?
Nitrogen oxide (NO2). A highly poisonous gas. Exposure produces inflammation of lungs that may only cause slight pain or pass unnoticed, but resulting edema several days later may cause death.Is no2 acidic or basic?
Nitrogen dioxide is an acidic gas and produce an acidic solution in the water (mixture of acids). Otherwise, we can say, NO2 is one of the strong acidic gas in chemistry. Nitrogen dioxide is an acidic gas. NO2 dissolves very well in water react with water to give nitrous acid and nitric acid.Why is nitrogen dioxide dangerous?
The main effect of breathing in raised levels of nitrogen dioxide is the increased likelihood of respiratory problems. Nitrogen dioxide inflames the lining of the lungs, and it can reduce immunity to lung infections. This can cause problems such as wheezing, coughing, colds, flu and bronchitis.What does nitrogen dioxide smell like?
Nitrogen oxides, NO and NO2 as NO2. Oxides of nitrogen (NOx) is the term usually used to refer to nitrogen monoxide (nitric oxide or NO; a colourless and odourless gas) and nitrogen dioxide (NO2; a red-brown gas with a pungent smell which is soluble in water).Is nitrogen dioxide heavier than air?
Nitric oxide is rapidly oxidized in air at high concentrations to form nitrogen dioxide. exposures. Nitrogen dioxide is heavier than air, such that exposure in poorly ventilated, enclosed, or low-lying areas can result in asphyxiation. Both nitrogen dioxide and nitric oxide are gases at room temperature.What is nitrogen dioxide used for?
Uses: Nitrogen Dioxide, NO 2 , has been used as a catalyst in certain oxidation reactions; as an inhibitor to prevent polymerization of acrylates during distillation; as a nitrating agent for organic compounds; as an oxidizing agent; as a rocket fuel; as a flour bleaching agent and in increasing the wet strength ofIs no2 flammable?
Nitric oxide (NO) and nitrogen dioxide (NO2) are two gases whose molecules are made of nitrogen and oxygen atoms. Nitric oxide is a colorless, flammable gas with a slight odor. Nitrogen dioxide is a deep red-orange gas that is poisonous but not flammable.When nitrogen dioxide is dissolved in water it forms?
When nitrogen dioxide reacts with water, Mono-nitrogen oxides eventually form nitric acid when dissolved in atmospheric moisture, forming a component of acid rain. Nitrogen dioxide is an non-metallic oxide which is acidic in nature . Nitrogen dioxide is acidic in nature because it form acid when added in water.What is the Colour of nitrogen dioxide?
orange
What is nitrogen dioxide made of?
Nitrogen dioxide is an irritant gas, which at high concentrations causes inflammation of the airways. When nitrogen is released during fuel combustion it combines with oxygen atoms to create nitric oxide (NO). This further combines with oxygen to create nitrogen dioxide (NO2).How much nitrogen dioxide is dangerous?
Exposure above 150 ppm for 30 min to an hour results in fatal pulmonary edema or asphyxia and can result in rapid death (Lowry and Schuman 1956; NRC 1977; Mayorga 1994).What is water soluble nitrogen?
Water Soluble Nitrogen Soluble N fertilizers that contain ammonium N include urea, ammonium sulfate, and ammonium chloride. These fertilizers can produce excellent quality turf without leaching or runoff problems if used properly. The ammonium N can be absorbed by the soil, reducing the potential for N movement.Why is nitrogen not soluble in water?
Originally Answered: Why does oxygen and nitrogen are less soluble in water? It is because both these gases are non-polar , water is a polar solvent and so non-polar species will be insoluble in it!