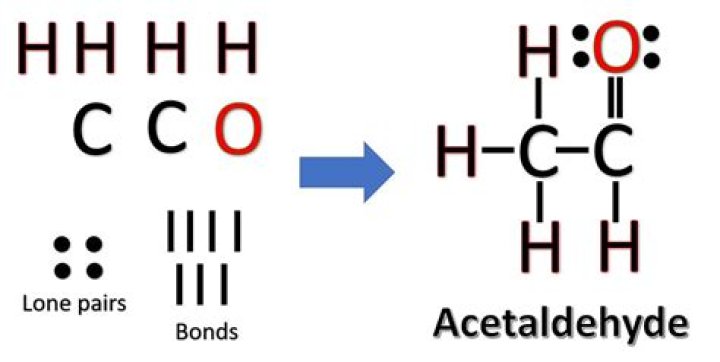
.
People also ask, what is the central atom in a Lewis structure?
The central atom is usually the least electronegative element in the molecule or ion; hydrogen and the halogens are usually terminal. Determine the total number of valence electrons in the molecule or ion. Add together the valence electrons from each atom.
One may also ask, how do you know which atom is central? Method 1: Compare Electronegativity The electronegativity of an element is its propensity to attract electrons, and the element in a compound with the lowest electronegativity is usually the central one. The exception to this rule is hydrogen, which is never the central atom except in the H2 molecule.
Likewise, what are the rules for drawing Lewis dot structures?
General rules for drawing Lewis structure
- Count up the total number of valence electrons.
- Calculate the total number of electrons that would be needed for each atom to have an octet (or doublet for H).
- Subtract the result of step 1 from the result of step 2.
- Assign two bonding electrons to each bond.
What is Lewis rule?
Lewis Dot Rules. In 1916, ten years before the Schrodinger wave equation, G. N. Lewis suggested that a chemical bond involved sharing of electrons. He described what he called the cubical atom , because a cube has 8 corners, to represent the outer valence shell electrons which can be shared to create a bond.
Related Question AnswersWhat is the Lewis structure for NOCl?
In the NOCl Lewis structure Nitrogen (N) is the least electronegative atom and goes in the center of the Lewis structure. Check the formal charges to be sure that each atom has a formal charge of zero. In the Lewis structure for NOCl there are a total of 18 valence electrons.What are the two main types of chemical bonds?
There are two main types of chemical bonds that hold atoms together: covalent and ionic/electrovalent bonds. Atoms that share electrons in a chemical bond have covalent bonds. An oxygen molecule (O2) is a good example of a molecule with a covalent bond.How do you determine formal charge?
The formal charge is determined by the valence electrons (VE), nonbonding electrons (NE), and bonding electrons (BE) in the following formula: By calculating the formal charge of each atom in a molecule, we can determine the overall charge of the molecule.How do you determine hybridization?
A Shortcut For Determining The Hybridization Of An Atom In A Molecule- Look at the atom.
- Count the number of atoms connected to it (atoms – not bonds!)
- Count the number of lone pairs attached to it.
- Add these two numbers together.
Why is carbon always the central atom?
Consider the bonding patterns of our component atoms: Carbon is always a central atom because it forms four bonds. Oxygen tends to form two bonds and two lone pairs. Hydrogen atoms form one bond. The total number of valence electrons the atoms brought into the molecule is 12.What is the most electronegative element?
Electronegativity varies in a predictable way across the periodic table. Electronegativity increases from bottom to top in groups, and increases from left to right across periods. Thus, fluorine is the most electronegative element, while francium is one of the least electronegative.Why is hydrogen never the central atom?
Hydrogen can't be the central atom because it can only form one bond, so it must generally be in the outer layer of atoms. When drawing a lewis structure, it's recommended for hydrogen atoms to be added in last.What is the least electronegative atom?
Most and Least Electronegative Elements The most electronegative element on the periodic table is fluorine (3.98). The least electronegative element is cesium (0.79). The opposite of electronegativity is electropositivity, so you could simply say cesium is the most electropositive element.Why is the least electronegative the central atom?
The least electronegative elements in the center of lewis structures because an atom in the central position shares more of its electrons than does a terminal atom. Atoms with higher electronegative are generally more reluctant to share its electrons.What does a Lewis structure not show?
Lewis structure does NOT attempt to explain the geometry of molecules, how the bonds form, or how the electrons are shared between the atoms. It is the simplest and most limited theory on electronic structure.What is the role of the central atom when drawing a Lewis structure?
What is the role of the central atom when drawing the Lewis structure for a molecule? All terminal atoms are bonded to it. Atoms lose, gain, or share electrons to end with a full outer energy level. Certain atoms share electrons to achieve an octet.What are the exceptions to the octet rule?
However, there are three general exceptions to the octet rule: Molecules, such as NO, with an odd number of electrons; Molecules in which one or more atoms possess more than eight electrons, such as SF 6; and. Molecules such as BCl 3, in which one or more atoms possess less than eight electrons.How do you determine polarity?
Step 2: Identify each bond as either polar or nonpolar. (If the difference in electronegativity for the atoms in a bond is greater than 0.4, we consider the bond polar. If the difference in electronegativity is less than 0.4, the bond is essentially nonpolar.) If there are no polar bonds, the molecule is nonpolar.Which element has the highest ionization energy?
From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).How do you find the bond order?
If there are more than two atoms in the molecule, follow these steps to determine the bond order:- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.