Covalent bonds can be single, double, and triple bonds. Single bonds occur when two electrons are shared and are composed of one sigma bond between the two atoms. Double bonds occur when four electrons are shared between the two atoms and consist of one sigma bond and one pi bond..
Regarding this, which electrons are shared in covalent bonds?
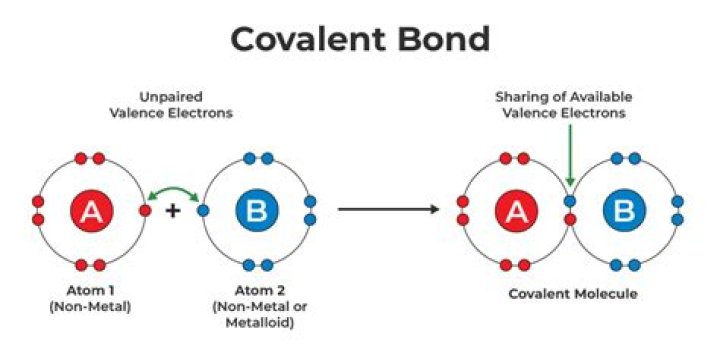
A covalent bond, also called a molecular bond, is a chemical bond that involves the sharing of electron pairs between atoms. These electron pairs are known as shared pairs or bonding pairs, and the stable balance of attractive and repulsive forces between atoms, when they share electrons, is known as covalent bonding.
Additionally, is electrons shared in covalent compounds? Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability.
Hereof, what is the total number of electrons shared in a double covalent bond?
In a covalent bond a pair of electrons are shared between the two atoms 'connected' by the covalent bond. So a double covalent bond has two pairs of electrons being shared, so in total four electrons.
What kind of bond is made up of shared electrons?
An ionic bond is a type of chemical bond formed through an electrostatic attraction between two oppositely charged ions. Ionic bonds are formed between a cation, which is usually a metal, and an anion, which is usually a nonmetal. A covalent bond involves a pair of electrons being shared between atoms.
Related Question Answers
Which covalent bond is strongest?
sigma bond
How do you know how many electrons are shared in a covalent bond?
Oxygen can also form covalent bonds, however, it needs a further 2 electrons to complete its valence shell (it has 6). Two oxygen atoms must share 2 electrons each to complete each other's shells, making a total of 4 shared electrons. Because twice as many electrons are shared, this is called a 'double covalent bond'.Is h2o a covalent bond?
A water molecule, abbreviated as H2O, is an example of a polar covalent bond. The electrons are unequally shared, with the oxygen atom spending more time with electrons than the hydrogen atoms. Since electrons spend more time with the oxygen atom, it carries a partial negative charge.How many electrons are shared in a single covalent bond?
two electrons
What are the properties of two atoms in a covalent bond?
What property of the two atoms in a covalent bond determines whether or not the bond will be polar? The difference between the electronegativity of the two atoms in a bond will determine whether the bond is ionic or covalent. If the difference in electronegativity is greater than 1.7, the bond is considered ionic.What is covalent bond and example?
Covalent Bond Examples Each of the covalent bonds contains two electrons, one from a hydrogen atom and one from the oxygen atom. Both atoms share the electrons. A hydrogen molecule, H2, consists of two hydrogen atoms joined by a covalent bond.Is NaCl a covalent bond?
Ionic bonds usually occur between metal and nonmetal ions. For example, sodium (Na), a metal, and chloride (Cl), a nonmetal, form an ionic bond to make NaCl. In a covalent bond, the atoms bond by sharing electrons. Covalent bonds usually occur between nonmetals.What is the structure of a covalent bond?
The attraction for the shared pair of electrons is a covalent bond. This force holds the atoms together in a molecule. Shared pair of electrons spend time orbiting oxygen to give it 8 valence electrons and orbiting hydrogen to give them 2 valence electrons.How many total electrons are shared in a triple covalent bond?
One covalent bond contains 2 electrons, one from each atom. A triple bond indicates that, well, there are three total covalent bonds in that one bond. So, 3 bonds x 2 electrons per bond would give you 6 total electrons.How many electrons are being shared in a quadruple covalent bond?
eight
How many electrons does a bond represent?
A covalent bond is pictured as two electrons shared by two different atomic nuclei, which means that each of the two nuclei has two electrons.How many electrons are in a triple bond?
A triple bond in chemistry is a chemical bond between two atoms involving six bonding electrons instead of the usual two in a covalent single bond. The most common triple bond, that between two carbon atoms, can be found in alkynes.What is the weakest bond?
A Chemical bond is technically a bond between two atoms that results in the formation of a molecule , unit formula or polyatomic ion. The weakest of the intramolecular bonds or chemical bonds is the ionic bond. The ionic bond is generally the weakest of the true chemical bonds that bind atoms to atoms.How many electrons are shared in a double covalent bond quizlet?
A double covalent bond is where two pairs of electrons are shared between the atoms rather than just one pair.How many individual electrons are located in a double bond?
In a single bond one pair of electrons is shared, with one electron being contributed from each of the atoms. Double bonds share two pairs of electrons and triple bonds share three pairs of electrons.Which bond is most polar?
The quick answer - right from the get-go, since nitrogen is one of the most electronegative elements in the periodic table, the bond it forms with hydrogen will be the most polar out of all those listed. The polarity of a bond is given by the difference in electronegativity between the two atoms that form said bond.How coordinate bond is formed?
A coordinate bond is formed when a ligand molecule (or ion) that has extra lone pairs on it uses those pairs to join another atom that has empty valence orbitals. The bond that is formed is the lone pair sharing into the empty orbital of the second atom.Why is water a polar molecule?
A water molecule, because of its shape, is a polar molecule. That is, it has one side that is positively charged and one side that is negatively charged. The molecule is made up of two hydrogen atoms and one oxygen atom. The bonds between the atoms are called covalent bonds, because the atoms share electrons.What are 3 types of covalent bonds?
The three types as mentioned in the other answers are polar covalent, nonpolar covalent, and coordinate covalent. The first, polar covalent, is formed between two nonmetals that have a difference in electronegativity. They share their electron density unevenly.