.
Also asked, what does GMP mean in the food industry?
good manufacturing practices
Also, what are the 5 main components of good manufacturing practice? To simplify this, GMP helps to ensure the consistent quality and safety of products by focusing attention on five key elements, which are often referred to as the 5 P's of GMP—people, premises, processes, products and procedures (or paperwork). And if all five are done well, there is a sixth P … profit!
Thereof, why GMP is important in food industry?
GMP is important to ensure that businesses produce safe food to the public. Businesses in the food industry have a legal and moral responsibility to prepare food that is safe for the consumer. By not implementing adequate good manufacturing practices (GMP), a food business can risk several negative consequences.
What is a GMP policy?
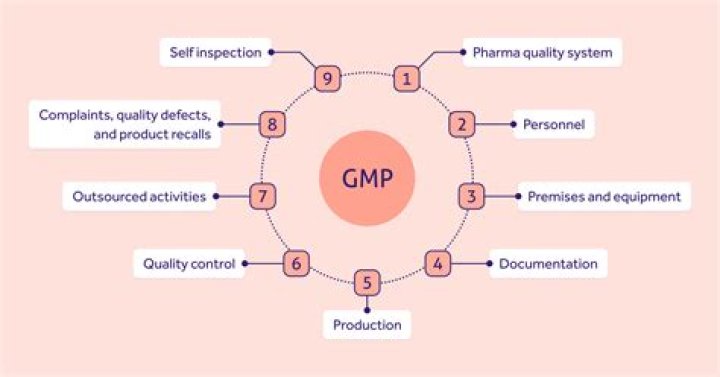
GMP Policy. Good Manufacturing Practice (GMP) is a set of regulations and guidelines that provide guidance concerning the manufacturing, holding and distri- bution of active pharmaceutical ingredients (APIs), chemical intermediates used to produce APIs, medicinal (drug) products, food and food/feed additives.
Related Question AnswersWhat are the principles of GMP?
A basic outline of GMP All guidelines follow a few basic principles: The production and distribution of the drugs must minimize any risk to their quality. Manufacturing facilities must maintain a clean and hygienic manufacturing area, including laboratories and storage.What is GMP and GHP?
O PRPs (PRE-REQUISITE PROGRAM) are also referred to as good hygienic practices(GHP), good agricultural practices, good production practices, good manufacturing practices(GMP), good distribution practices, and good trading practices.What is a GMP audit?
A GMP audit is a check to see if your food business and food handlers are complying with the required good manufacturing practices.What is the importance of GMP?
What is GMP and why is it so important? Good Manufacturing Practices (GMPs) are systems created and mandated by the government to regulate production, verification and validation of drugs, food and/or medical devices, ensuring that finished products are effective and safe for market distribution.What is the purpose of GMP?
Good Manufacturing Practice (GMP) is a system for ensuring that products are consistently produced and controlled according to quality standards. It is designed to minimize the risks involved in any pharmaceutical production that cannot be eliminated through testing the final product.What is the difference between Haccp and GMP?
GMP (Good Manufacturing Practice) is a system to ensure that products meet food safety, quality and legal requirements. As a food manufacturer you should have GMP in place. HACCP (Hazard Analysis and Critical Control Point) can be part of GMP and is a systematic program to assure food safety.What is a GMP warehouse?
The warehouse plays a crucial role in a medicinal products factory. This module explains the requirements of Good Manufacturing Practice (GMP) for the warehouse, and how to comply with them. GMP for the warehouse overlaps with Good Distribution Practice (GDP), which applies to the whole distribution chain for products.Does FDA issue GMP certificates?
Do anybody know why US FDA do not issues a GMP certificate or any form of certificate to a medical device manufacturer after the inspection? No certificate issued - FDA would give you Establishment Inspection Report.What is good manufacturing practices in food industry?
Good Manufacturing Practices* (GMPs) are the basic operational and environmental conditions required to produce safe foods. They ensure that ingredients, products and packaging materials are handled safely and that food products are processed in a suitable environment.How do I write a GMP?
Read on to find out my five steps to writing an effective good manufacturing practices program for your food business.- What is a Good Manufacturing Practices Program?
- Components of a GMP Program.
- How to write your GMP program.
- Step 1: Research.
- Step 2: Document.
- Step 3: Implement.
- Step 4: Record.
- Step 5: Review.
What is safe food?
Food Safety refers to handling, preparing and storing food in a way to best reduce the risk of individuals becoming sick from foodborne illnesses. The principles of food safety aim to prevent food from becoming contaminated and causing food poisoning.What are the current good manufacturing practices?
In the United States, Current Good Manufacturing Practices (cGMP) are the Food and Drug Administration's (FDA) formal regulations regarding the design, monitoring, control, and maintenance of manufacturing processes and facilities.What is GMP PDF?
? DEFINITION: GMP is that part of Quality Assurance which ensures that products are. consistently produced and controlled to the quality standard appropriate to their intended. use and as required by the marketing authorization.How do I become GMP certified?
Acquiring GMP Certification The application for GMP certification has to be made by an authorized person within the company seeking the certification. This is usually one with responsibility such as a Production Manager, a Quality Assurance Manager, a Quality Control Manager or the Managing Director.What are the 6 Quality Systems?
The six systems referred to in this inspection model are: quality, production, facilities and equipment, laboratory controls, materials, and packaging and labeling.What is the role of GMP?
What is GMP? Good manufacturing practice (GMP) is a system for ensuring that products are consistently produced and controlled according to quality standards. It is designed to minimize the risks involved in any pharmaceutical production that cannot be eliminated through testing the final product.What are the 4 types of quality control?
There are seven primary quality control tools which include:- Checklists. At its most basic, quality control requires you to check off a list of items that are imperative to manufacture and sell your product.
- Fishbone diagram.
- Control chart.
- Stratification.
- Pareto chart.
- Histogram.
- Scatter Diagram.