.
Also question is, when an electron jumps to a higher energy level?
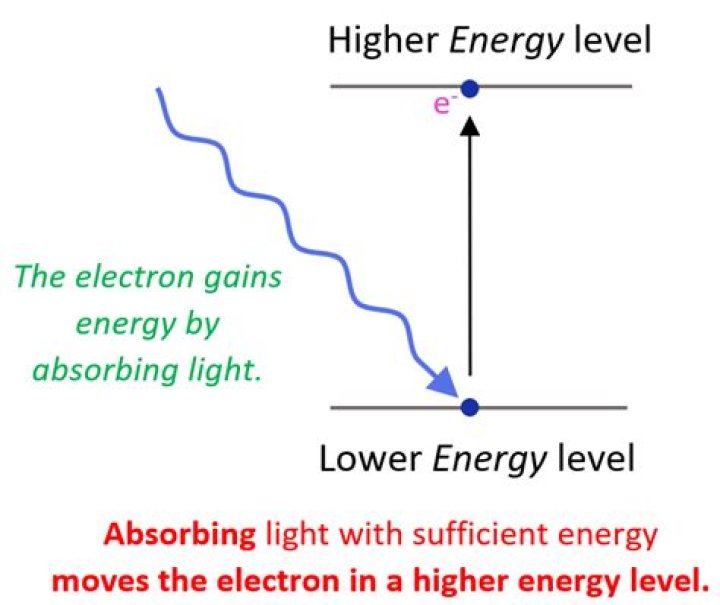
The electron can gain the energy it needs by absorbing light. If the electron jumps from the second energy level down to the first energy level, it must give off some energy by emitting light. The atom absorbs or emits light in discrete packets called photons, and each photon has a definite energy.
Furthermore, what happens when an electron jumps from energy level 1 to energy level 4 in an atom? Question: Case 1: In An Atom, An Electron Jumps From Energy Level 4 To Energy Level 2. During This Process, O A Proton Was Absorbed By The Atom. O A Photon Was Emitted By The Atom. O A Proton Was Emitted By The Atom.
Furthermore, what happens when electron jumps from lower energy level to higher energy level?
When an electron jumps from a lower energy level to a higher energy level, it gains energy. Conversely, when an electron jumps from a higher energy level to a lower energy level, it loses energy, which is given out in the form of radiation.
What happens when an electron jumps from energy level 4 to energy level 2 in an atom?
When electrons move from the excited to the ground state, energy is emitted. From the problem given, the electron jumps from energy level 4 to 2. It has moved from a higher energy level to a lower one and energy is emitted. The energy emitted must be commensurate with that of the new lower energy level.
Related Question AnswersWhat happens when an atom emits energy?
How do atoms emit light? Light is the result of electrons moving between defined energy levels in an atom, called shells. When something excites an atom, such as a collision with another atom or a chemical electron, an electron may absorb the energy, boosting it up to a higher-level shell.What happens when an atom absorbs energy?
When an electron absorbs energy, it jumps to a higher orbital. An electron in an excited state can release energy and 'fall' to a lower state. When it does, the electron releases a photon of electromagnetic energy. The electron can absorb one quantum of energy and jump up to the excitation state.What happens when an electron jumps from energy level 3 to energy level 7 in an atom?
Case 1: In an atom, an electron jumps from energy level 3 to energy level 7. During this process, a photon was emitted by the atom a photon was absorbed by the atom. a proton was absorbed by the atom. a proton was emitted by the atom.Can electrons absorb any amount of energy?
An atom changes from a ground state to an excited state by taking on energy from its surroundings in a process called absorption. The electron absorbs the energy and jumps to a higher energy level. In the reverse process, emission, the electron returns to the ground state by releasing the extra energy it absorbed.When an electron jumps from one position to another?
Atomic electron transition is a change of an electron from one energy level to another within an atom or artificial atom. It appears discontinuous as the electron "jumps" from one energy level to another, typically in a few nanoseconds or less.Why is there so much energy in an atom?
Because the potential energy keeping an atom together is so strong. The energy keeping the nucleus together is strong. An atom includes both the nucleus and electrons, and the binding energy of electrons is magnitudes smaller than nuclear binding energy. So atoms have something called "nuclear binding energy".What is the energy level of an electron?
Energy levels inside an atom are the specific energies that electrons can have when occupying specific orbitals. Electrons can be excited to higher energy levels by absorbing energy from the surroundings. Light is emitted when an electron relaxes from a high energy state to a lower one.How do you determine energy levels?
Number of energy levels in each period- The atoms in the first period have electrons in 1 energy level.
- The atoms in the second period have electrons in 2 energy levels.
- The atoms in the third period have electrons in 3 energy levels.
- The atoms in the fourth period have electrons in 4 energy levels.
Where do electrons get their energy from?
It requires an external source of energy in order to change to a higher-energy state or else to leave the atom and become a free electron again. Typically it gets this energy either by absorbing a photon or by absorbing some kinetic energy when its atom hits another atom.Which energy level has the least energy?
At the lowest energy level, the one closest to the atomic center, there is a single 1s orbital that can hold 2 electrons. At the next energy level, there are four orbitals; a 2s, 2p1, 2p2, and a 2p3. Each of these orbitals can hold 2 electrons, so a total of 8 electrons can be found at this level of energy.Which of the following energy level changes for an electron is least energetic?
33 Cards in this Set| what is the subatomic particle having a negligible mass and a negative charge? | electron |
|---|---|
| which of the following energy-level changes for an electron is least energetic? a. 2-1 b. 5-4 c. 3-2 d. 4-3 e. all changes have the same energy | b. 5-4 |
| what is the term for a particle of radiant energy? | photon |
How many electrons can each energy level hold?
Each shell can contain only a fixed number of electrons: The first shell can hold up to two electrons, the second shell can hold up to eight (2 + 6) electrons, the third shell can hold up to 18 (2 + 6 + 10) and so on. The general formula is that the nth shell can in principle hold up to 2(n2) electrons.Why can't electrons exist in between energy levels?
When an electron is hit by electricity, it can gain energy and can be bumped up to a higher energy orbit further away from the nucleus. On the other hand, when an electron loses energy, it falls back down to a lower energy orbit closer to the nucleus. The electron can never exist at distances in between allowed orbits.Why are energy levels discrete?
To jump from one energy level to a higher energy level the electron has to absorb light, meaning energy, it has to absorb a photon. It can not absorb part of a photon, therefore it can not have an in-between level, the levels are discrete, meaning there can be no in-between levels.How do you find the energy of an electron?
A simple expression for the energy of an electron in the hydrogen atom is:- E=−13.6n2 where the energy is in electron volts.
- n is the principle quantum number.
- So for an electron in n=1 :
- E=−13.6eV.
- To convert to joules you can x this by 1.6×10−19.