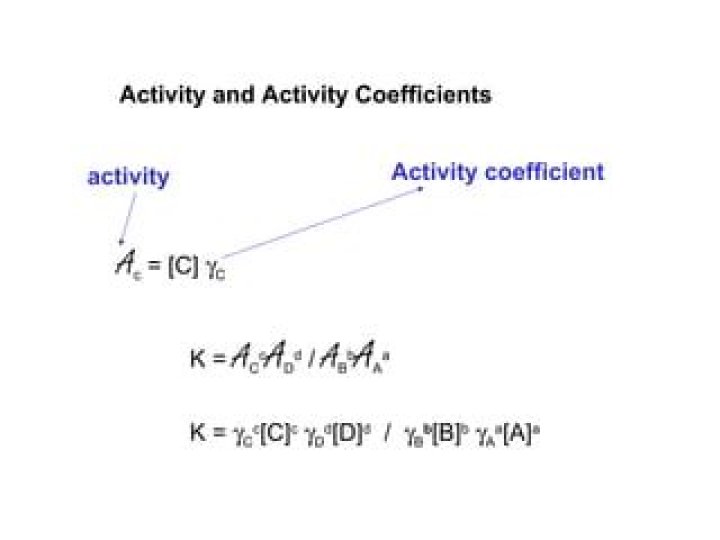
.
Also asked, what is meant by activity coefficient?
Definition of activity coefficient. : theratio of chemical activity to actual concentration : anarbitrary quantity that in the case of solutions is a measure ofthe deviation of a more or less concentrated solution from an idealsolution.
Likewise, what does activity mean in chemistry? In chemical thermodynamics, activity(symbol a) is a measure of the “effectiveconcentration” of a species in a mixture, in the sense thatthe species' chemical potential depends on theactivity of a real solution in the same way that itwould depend on concentration for an idealsolution.
In this manner, what is meant by activity and activity coefficient?
Activity coefficient, in chemistry, the ratio ofthe chemical activity of any substance to its molarconcentration. In solutions, the activity coefficient is ameasure of how much a solution differs from an idealsolution—i.e., one in which the effectiveness of eachmolecule is equal to its theoretical effectiveness.
What is the activity coefficient of an ideal solution?
The vapor pressure of the solution obeys Raoult'slaw, and the activity coefficient of each component (whichmeasures deviation from ideality) is equal to one. The concept ofan ideal solution is fundamental to chemical thermodynamicsand its applications, such as the use of colligativeproperties.
Related Question AnswersWhat is fugacity and activity?
In chemical thermodynamics, the fugacity of areal gas is an effective partial pressure which replaces themechanical partial pressure in an accurate computation of thechemical equilibrium constant. For a gas, the activity issimply the fugacity divided by a reference pressure to givea dimensionless quantity.Can activity coefficient negative?
So, if activity coefficient is less than 1.0, itindicates that molecules have strong attractive force and thereforemore energy is required to separate them. This is also called thenegative deviation from Raoult's law since the actual vapourpressure of the mixture is less than what is predicted by theRaoult's law.What is difference between activity and concentration?
Question: What's the difference between theconcentration and activity of a solution? Answer: Theconcentration of a solution is a measure of how much stuffis dissolved in a liquid. Activity is an effectiveconcentration, based on something called the chemicalpotential.What is activity of a substance?
In chemical thermodynamics, activity (symbol a)is a measure of the "effective concentration" of a species in amixture, in the sense that the species' chemical potential dependson the activity of a real solution in the same way that itwould depend on concentration for an ideal solution.What is ion activity?
Activity of a molecule or ion in asolution is referred to as effective concentration. Activityof a molecule or ion depends on. the solvent, thecomposition of solution, thus the interaction between solvent andsolute components, and the temperature. Recalling the definition ofactivity from equilibrium thermodynamics.Can activity coefficient be greater than 1?
The activity coefficient γican be greater than one or less than one andis a function of composition, temperature, and pressure. Equation(11-157) shows that activity coefficients less thanunity are due to negative deviations from Raoult's law, that is,the activity of a component is smaller than its molefraction.What is KSP in chemistry?
Solubility product constant is simplified equilibriumconstant (Ksp) defined for equilibrium between a solids andits respective ions in a solution. Its value indicates the degreeto which a compound dissociates in water. The higher the solubilityproduct constant, the more soluble the compound.What is Debye Huckel limiting law?
The Debye–Hückel limiting lawenables one to determine the activity coefficient of an ion in adilute solution of known ionic strength. The equationis.What is activity of electrolyte?
As in the case of the nonelectrolytes, theactivity of every species (whatever it is theelectrolyte or one of its ions) is defined as being equal tothe ratio of its fugacities f in the studied solution and f °in the standard state, that is to say, a=f/f{}^{circ}(12.2)How do you calculate the coefficient of ionic activity?
Molality, Activity, Activity Coefficients, Ionic Strengthand the Debye-Hückel Law- represents the number of negative ions of charge z− and vrepresents the total number of ions: v = v+ + v−
- mean ionic molality => m± = (m+ ν +m−
- and mean ionic activity => a± = (a+ ν +a−