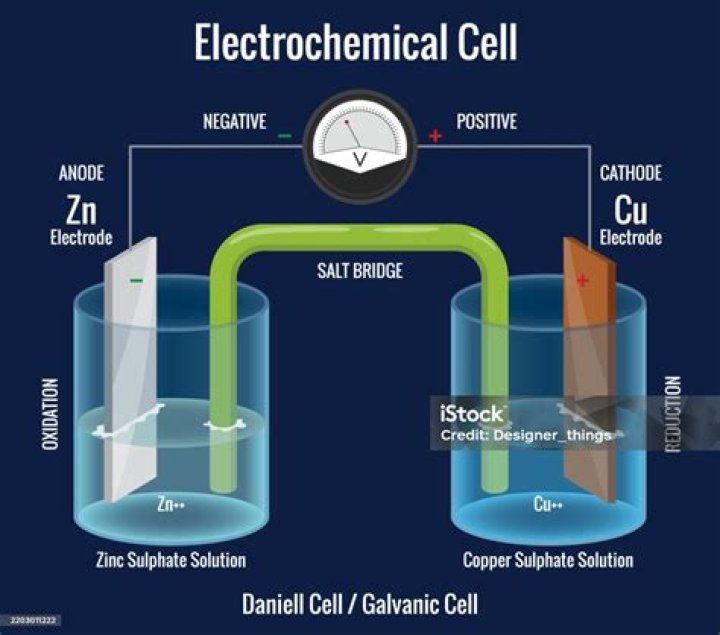
An electrochemical cell is a device capable of either generating electrical energy from chemical reactions or using electrical energy to cause chemical reactions..
Similarly, it is asked, what is an electrochemical cell and how does it work?
Electrochemical cells use chemical reactions to generate electricity or electricity to energize chemical reactions. There are two types: electrochemical cells use an applied source of energy to produce a chemical reaction; galvanic cells use a chemical reaction, usually a redox reaction, to produce electricity.
Subsequently, question is, what are the two types of electrochemical cells? Two Types of Cell There are two fundamental types of electrochemical cell: galvanic and electrolytic. Galvanic cells convert chemical potential energy into electrical energy. The energy conversion is achieved by spontaneous (ΔG < 0) redox reactions producing a flow of electrons.
In this manner, what is electrochemical cell example?
A common example of an electrochemical cell is a standard 1.5-volt cell which is used to power many electrical appliances such as TV remotes and clocks. Such cells capable of generating an electric current from the chemical reactions occurring in them care called Galvanic cells or Voltaic cells.
What are the main components of an electrochemical cell?
The electrochemical cell consists of four main parts:
- The anode: the compartment where oxidation occurs.
- The cathode: the compartment where reduction occurs.
- External pathway to allow the flow of electrons.
- Salt bridge or porous barrier: allows ions to flow back and forth so that charge does not build up.
Related Question Answers
What is electrochemical process?
Electrochemical reaction, any process either caused or accompanied by the passage of an electric current and involving in most cases the transfer of electrons between two substances—one a solid and the other a liquid.What are the two main components of an electrochemical cell?
Electrochemical cells have two conductive electrodes, called the anode and the cathode. The anode is defined as the electrode where oxidation occurs. The cathode is the electrode where reduction takes place.What are some useful applications of electrochemical cells?
Batteries have many
uses including in: A fuel
cell converts
the chemical potential energy produced by
the oxidation of fuels (e.g. hydrogen gas, hydrocarbons, alcohols) into electrical energy.
Electroplating (ESCRQ)
- corrosion protection.
- abrasion and wear resistance.
- the production of jewellery.
How do electrochemical cells work?
An electrochemical cell can be created by placing metallic electrodes into an electrolyte where a chemical reaction either uses or generates an electric current. Electrochemical cells which generate an electric current are called voltaic cells or galvanic cells, and common batteries consist of one or more such cells.Who invented electrochemical cell?
Alessandro Volta
Who discovered electrochemical cell?
Volta was the inventor of the voltaic pile, the first electrical battery. In common usage, the word "battery" has come to include a single galvanic cell, but a battery properly consists of multiple cells.What a cell is?
The cell (from Latin cella, meaning "small room") is the basic structural, functional, and biological unit of all known organisms. A cell is the smallest unit of life. Cells consist of cytoplasm enclosed within a membrane, which contains many biomolecules such as proteins and nucleic acids.What is cell reaction?
The cell reaction is the overall reaction which takes place in the cell, written on the assumption that the right hand electrode is the cathode, i.e. assuming that the spontaneous reaction is the one in which reduction is occurring in the right-hand compartment. If it is, the reaction is spontaneous as written.What is a cell in physics?
A cell is a single electrical energy source which uses chemical reactions to produce a current*. The symbol for a cell is: Electricity Menu.What is anode and cathode?
Definition: The anode of a device is the terminal where current flows in from outside. The cathode of a device is the terminal where current flows out. By current we mean the positive conventional current. Since electrons are negatively charged, positive current flowing in is the same as electrons flowing out.Where are voltaic cells used?
Voltaic cells are typically used as a source of electrical power. By their nature, they produce direct current. A battery is a set of voltaic cells that are connected in parallel. For instance, a lead–acid battery has cells with the anodes composed of lead and cathodes composed of lead dioxide.What is Daniell cell used for?
The Daniell cell can be used to 'generate' electricity, by consuming an electrode, or to store electricity.What is primary cell give an example?
An electric cell that produces current by an irreversible chemical reaction is a primary cell. Primary batteries are “single use” and cannot be recharged. Dry cells and (most) alkaline batteries are examples of primary batteries. The second type is rechargeable and is called a secondary battery.How does a cell produce electricity?
Answer: In simple cell battery converts chemical energy into electricity. The chemical reaction produces electrons, which then collect at the negative terminal of the battery. When there is a connection between the positive and negative terminal, the electrons flow to the positive terminal.Is anode positive or negative?
In a galvanic (voltaic) cell, the anode is considered negative and the cathode is considered positive. This seems reasonable as the anode is the source of electrons and cathode is where the electrons flow. However, in an electrolytic cell, the anode is taken to be positive while the cathode is now negative.Is it cathode minus anode?
The first equation is correct. The cathode is actually the reduction step, and the anode is the oxidation step. Interestingly, cathodes are red and are positive. So I have found many uses for this along the way.What is salt bridge in chemistry?
A salt bridge, in electrochemistry, is a laboratory device used to connect the oxidation and reduction half-cells of a galvanic cell (voltaic cell), a type of electrochemical cell. It maintains electrical neutrality within the internal circuit, preventing the cell from rapidly running its reaction to equilibrium.What is EMF of a cell?
The electromotive force (e) or e.m.f. is the energy provided by a cell or battery per coulomb of charge passing through it, it is measured in volts (V). It is equal to the potential difference across the terminals of the cell when no current is flowing.What is cell in chemistry?
A chemical cell converts chemical energy into electrical energy. Most batteries are chemical cells. A chemical reaction takes place inside the battery and causes electric current to flow. Batteries come in many shapes and sizes, from very small ones used in toys and cameras, to those used in cars or even larger ones.