.
Similarly, it is asked, how do you find the oxidation number?
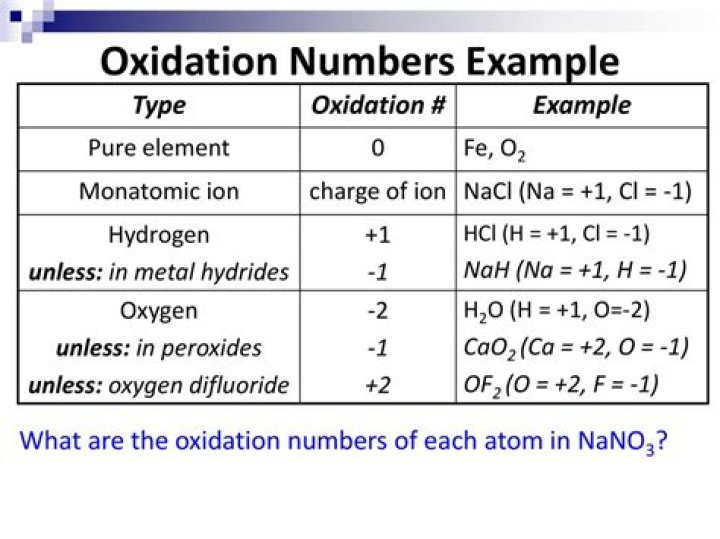
The oxidation number of a monatomic ion equals the charge of the ion. The oxidation number of H is +1, but it is -1 in when combined with less electronegative elements. The oxidation number of O in compounds is usually -2, but it is -1 in peroxides. The oxidation number of a Group 1 element in a compound is +1.
Likewise, what is mean by oxidation state? Definition of oxidation state. : a positive or negative number that represents the effective charge of an atom or element and that indicates the extent or possibility of its oxidation the usual oxidation state of sodium is +1 and of oxygen −2.
Also question is, what is Lithium's oxidation number?
So, the oxidation number for lithium is +1 because it is a metal that belongs in Group 1, and the oxidation number of magnesium is +2 because it is a metal that belongs in Group 2.
What is the oxidation number for Group 1?
Since Group 1 metals always have an oxidation state of +1 in their compounds, it follows that the hydrogen must have an oxidation state of -1 (+1 -1 = 0).
Related Question AnswersIs oxidation state and Valency same?
1. Valency is the maximum no. of electrons which an atom can lose or gain while oxidation no. is the actual no. of electrons which the element lose or gain to form a molecule. 2. Valency is the property of an isolated atom while oxidation state is calculated for an atom is present in a molecule i.e, in a bonded state.What is the oxidation number of co2?
The oxidation number of C in carbon dioxide (CO2) is (rules 1 & 2): 0 + (2 x 2) = +4 [Check (rule 3): +4 + 2(-2) = 0] The oxidation number of C in methane (CH4) is (rules 1 & 2): 0 – (4 x1) = -4 [Check (rule 3): -4 + 4(-1) = 0].What are the rules of oxidation number?
Rules for Assigning Oxidation Numbers to Elements- Rule 1: The oxidation number of an element in its free (uncombined) state is zero — for example, Al(s) or Zn(s).
- Rule 2: The oxidation number of a monatomic (one-atom) ion is the same as the charge on the ion, for example:
- Rule 3: The sum of all oxidation numbers in a neutral compound is zero.
What are oxidation numbers used for?
Oxidation numbers are used by chemists to keep track of electrons within a compound. We can use guidelines to assign oxidation numbers to atoms in a compound. Changes in oxidation state during a reaction tell us that there is a transfer of electrons.What is the oxidation number of o2?
The oxidation state of oxygen in its compounds is -2, except for peroxides like H2O2, and Na2O2, in which the oxidation state for O is -1. The oxidation state of hydrogen is +1 in its compounds, except for metal hydrides, such as NaH, LiH, etc., in which the oxidation state for H is -1.What is a good oxidizing agent?
This means that they easily attract electrons. As you can see in the periodic table of elements, the halogens that are good oxidizing agents are fluorine, chlorine, bromine and iodine, with fluorine being the strongest oxidizing agent among the four, followed by chlorine, bromine and iodine.What do you mean by Valency?
In chemistry, the valence or valency of an element is a measure of its combining power with other atoms when it forms chemical compounds or molecules. The concept of valence was developed in the second half of the 19th century and helped successfully explain the molecular structure of inorganic and organic compounds.Is oxidation positive or negative?
Oxidation occurs when the oxidation state of an atom, molecule, or ion becomes more positive. Reduction occurs when the oxidation state of an atom, molecule, or ion becomes more negative.Which element has highest oxidation number?
iridiumWhat is Oxidation Number Class 11?
Redox Reaction of Class 11 In ozonide the oxidation number of oxygen is assigned −1/3. In accordance with principle of conservation of charge, the algebric sum of the oxidation number of all the atoms in molecule is zero. But in case of polyatomic ion the sum of O.N. of all its atoms is equal to charge on the ion.How do you know which oxidation state is the highest?
To find the highest oxidation state in non-metals, from the number 8 subtract the number of the group in which the element is located, and the highest oxidation state with a plus sign will be equal to the number of electrons on the outer layer.What is the highest oxidation state for TC?
Both Tc and Re are reduced more readily in the lacunary α1 site as compared to the α2 site. The DFT calculations elucidate that the highest oxidation state attainable for Re is VII while, under the same electrochemistry conditions, the highest oxidation state for Tc is VI.What is the oxidation number of all elements?
Determining oxidation states| Element | Usual oxidation state | Exceptions |
|---|---|---|
| Group 2 metals | Always +2 | |
| Oxygen | Usually -2 | Peroxides and F2O (see below) |
| Hydrogen | Usually +1 | Metal hydrides (-1) (see below) |
| Fluorine | Always -1 |
Is oxidation state and oxidation number same?
Oxidation state and oxidation number are quantities that commonly equal the same value for atoms in a molecule and are often used interchangeably. Most of the time, it doesn't matter if the term oxidation state or oxidation number is used. Oxidation state refers to the degree of oxidation of an atom in a molecule.What is the oxidation number of copper?
The oxidation number of metallic copper is zero. In its compounds, the most common oxidation number of Cu is +2. Less common is +1. Copper can also have oxidation numbers of +3 and +4.How do you balance oxidation numbers?
Balancing Redox Reactions: Oxidation-Number-Change Method- Step 1: Assign oxidation numbers to each of the atoms in the equation and write the numbers above the atom.
- Step 2: Identify the atoms that are oxidized and those that are reduced.
- Step 3: Use a line to connect the atoms that are undergoing a change in oxidation number.
What are the free elements?
Free element. In chemistry, a free element is a chemical element that is not combined with or chemically bonded to other elements. Examples of elements which can occur as free elements include the oxygen molecule (O2) and carbon. All atoms of free elements have an oxidation number of 0.What is the oxidation number of P in h3po4?
We know from part (b) that the oxidation number of the phosphorus atoms in H3PO4 is +5.How do you find the oxidation number of h2so4?
Explanation:- In H2SO4 , hydrogen exists in its usual +1 state, and oxygen exists in its −2 state.
- Let x be the oxidation number of sulfur in sulfuric acid. Then we got:
- 1⋅2+x−2⋅4=0.
- 2+x−8=0.
- x−6=0.
- x=6.
- So, sulfur exists in the +6 state.