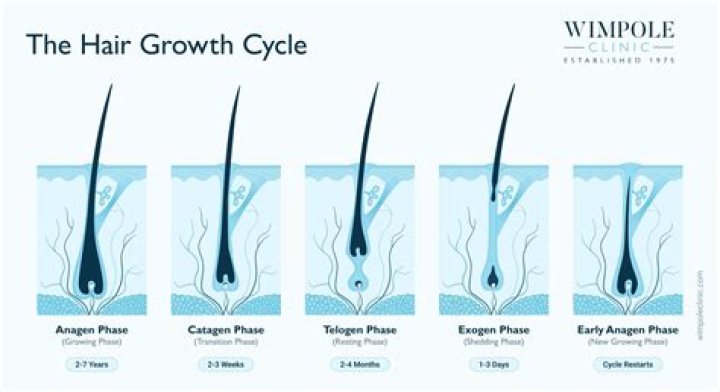
Hair is namely hygroscopic. This means that it absorbs water molecules from moist air and emits water molecules into dry air. This percentage is considered to be the normal moisture level of untreated hair..
Simply so, what makes something hygroscopic?
A hygroscopic substance is one that readily attracts water from its surroundings, through either absorption or adsorption. Examples include honey, glycerin, ethanol, methanol, concentrated sulfuric acid, and concentrated sodium hydroxide (lye).
Similarly, is Salt a hygroscopic? A. Unlike pepper, table salt is hygroscopic, meaning that because of the net positive charge of its chemical components, or ions, it can attract atmospheric water, which has a net negative charge. Traces of salt atop the shaker may attract visible water.
Also Know, what is the difference between Hydroscopic and hygroscopic?
Yes, hydroscopic is a word. It's an adjective derived from a noun: hydroscope. The word hydroscope is usually used to mean a device for observing underwater objects, say from a boat. The word hygroscopic is now more commonly used to describe a material that tends to absorb water.
How do you get moisture out of hair?
The more moisture for thick, coarse dry hair, the better. So use a rich, rinse-out conditioner after every shower, and follow it up with a moisturizing leave-in spray before styling. Once a week, apply a deep conditioning mask. Allow it to penetrate for five to 15 minutes for a silky, smooth result.
Related Question Answers
Is water hygroscopic?
Water is an important solvent, so it's unsurprising that there is a term specifically related to water absorption. A hygroscopic substance is able to absorb or adsorb water from its surroundings. Most hygroscopic materials are salts, but many other materials display the property.Is pure NaCl hygroscopic?
Hygroscopic nature means it tends to draw water or absorb water when exposed to air. pure sodium chloride does not attract water form just moist air. If the relatively humidity is low, the mineral halite will not be changed in decades.What are hygroscopic salts and give examples?
Examples of several known hygroscopic compounds are: Sodium chloride (halite) (NaCl). Calcium chloride (CaCl2). Sodium hydroxide (NaOH).Is baking soda hygroscopic?
Sugar is also hygroscopic; it forms weak bonds with the water molecules in its surroundings. Although baking soda is less hygroscopic, your cucumber slice with baking soda on it might still have lost some weight, which means that the baking powder is able to absorb some water from the cucumber.Why is NaOH hygroscopic?
Solid NaOH is very hygroscopic, that is it absorbs water from the air, so it is very hard to weigh exactly. This decreases the concentration of the OH- ions in the solution.Is oil a hygroscopic?
Oils by nature are hygroscopic, which means they absorb moisture from the air. The tendency of an oil to undergo this process is known as hygroscopicity.What material absorbs moisture?
3 Types of Moisture Absorbers Explained - Silica Gel. Silica Gel is a moisture adsorber made from sodium silicate.
- Clay Absorbers. Clay absorbers offer a cheap alternative to absorbing moisture in a home or in any other space where moisture leads to molds.
- Calcium Chloride. Perhaps the best moisture absorber is calcium chloride, a mixture of chlorine and calcium.
Is Honey hygroscopic?
Honey is, first and foremost, a sugar. Sugars are hygroscopic, a term that means they contain very little water in their natural state but can readily suck in moisture if left unsealed. But honey isn't the only hygroscopic food source out there.What does Hydroscopic mean?
Definition of hydroscope. : a device for enabling a person to see an object at a considerable distance below the surface of water by means of a series of mirrors enclosed in a steel tube — compare water glass.Is MgCl2 hygroscopic?
Magnesium Chloride is a colorless crystalline compound. The salt is very hygroscopic. The compound well dissolves in water with solubility of 35.5g per 100g H2O at 298 K. Dissolving MgCl2 is an exothermic process.Why is sugar hygroscopic?
Moistens: Sugar is hygroscopic, which means that water thinks it's pretty great—and really, who can blame it? If you ever put a lollipop in the fridge, only to return to a pool of bright pink sugar syrup, it is because of sugar's water-attracting property. But being hygroscopic does more than melt your lollies.What is Deliquescence?
chemistry. Deliquescence, the process by which a substance absorbs moisture from the atmosphere until it dissolves in the absorbed water and forms a solution. Deliquescence occurs when the vapour pressure of the solution that is formed is less than the partial pressure of water vapour in the air.Does water attract water?
Water is highly cohesive—it is the highest of the non-metallic liquids. More precisely, the positive and negative charges of the hydrogen and oxygen atoms that make up water molecules makes them attracted to each other.What is non hygroscopic material?
Non-hygroscopic plastics are. Polyethylene, Polypropylene, Polystyrene, PVC.Is wood a hygroscopic?
A fundamental fact is that wood is hygroscopic. This means that wood, almost like a sponge, will gain or lose moisture from the air based upon the conditions of the surrounding environment.Is HCL hygroscopic?
Hydrogen chloride is a colorless, corrosive, nonflammable gas with a pungent odor (Budavari et al. Hydrogen chloride is highly soluble in water and forms hydrochloric acid. Because hydrogen chloride is so hygroscopic, airborne hydrogen chloride is typically an aerosol of hydrochloric acid.Is Sugar hygroscopic?
Dry Ingredient with Wet Properties: Sugar Is Hygroscopic I mentioned that when certain materials get wet, they absorb the moisture and swell up. Sugar is one such material, but as you'll see, not all sugars are the same. Left: Granulated sugar, sucrose.Why does my salt get wet?
You see salt is hygroscopic which means that it naturally attracts water and that means if you leave it in any room where there is any humidity in the air then the air will stick to the salt lamps surface and eventually turn into water droplets which you see as leaking, or crying. Its totally normal.Does salt hydrate you?
The main difference is the amount of salt. When you're dehydrated, you need water, all right, but you need electrolytes (the scientific term for various salts) to survive too. Seawater has way too much of these salts, which can cause you to become more dehydrated than if you hadn't drunk anything at all.