So their solution is Emulsion. However, many hydraulic systems use a mixture of oil and water and that mixture is obtained by vigorous mechanical agitation and is called an emulsion (tiny droplets of the oil form a suspension in water)..
Similarly, it is asked, is a mixture of oil and water heterogeneous?
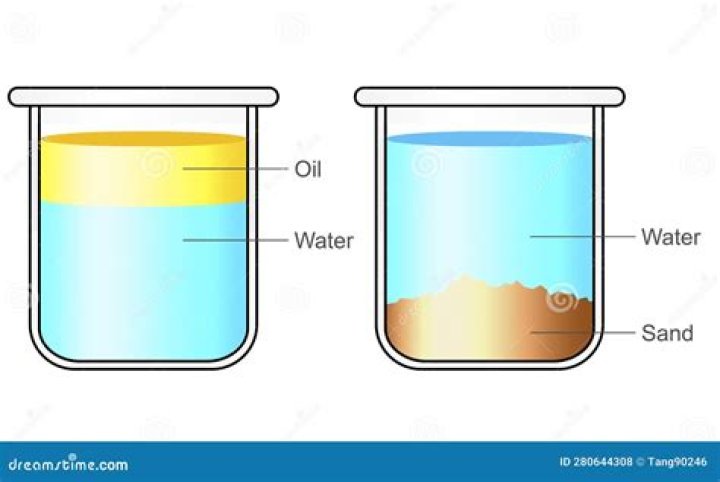
A solution of oil and water is a great example of a heterogeneous mixture. A solution of oil and water is a great example of a heterogeneous mixture. A homogeneous mixture is a mixture of two substances that is completely even throughout the solution and there are no borders inbetween substances.
Furthermore, how do you mix oil and water naturally? As a result, when you add oil to a cup of water the two don't mix with each other. Because oil is less dense than water, it will always float on top of water, creating a surface layer of oil.
Likewise, people ask, what is water and oil?
Why oil and water do not mix. Liquid water is held together by hydrogen bonds. Oils and fats not have any polar part and so for them to dissolve in water they would have to break some of water's hydrogen bonds. Water will not do this so the oil is forced to stay separate from the water.
What are 5 examples of heterogeneous mixtures?
Examples of Heterogeneous Mixtures
- Concrete is a heterogeneous mixture of an aggregate: cement, and water.
- Sugar and sand form a heterogeneous mixture.
- Ice cubes in cola form a heterogeneous mixture.
- Salt and pepper form a heterogeneous mixture.
- Chocolate chip cookies are a heterogeneous mixture.
Related Question Answers
What are the 2 types of mixture?
There are two types of mixture: (1) Homogenous mixture. (2) Heterogeneous mixture. A homogeneous mixture is defined as "the mixture, which has uniform composition through out its mass".Is baking soda a heterogeneous mixture?
Examples of pure substances include tin, sulfur, diamond, water, pure sugar (sucrose), table salt (sodium chloride) and baking soda (sodium bicarbonate). Depending on who you talk to, homogeneous mixtures may be considered examples of pure substances.Is blood a mixture?
Answer and Explanation: Blood is a mixture because it is made up of many different parts that are combined, but can be physically separated from each other.Is blood a heterogeneous mixture?
A glass of iced tea is a heterogeneous mixture. Blood is a heterogeneous mixture because the blood cells are physically separate from the blood plasma. The cells have different properties than the plasma. The cells can be separated from the plasma by centrifuging, which is a physical change.Is pizza a homogeneous mixture?
So pizza is not a compound. It is a mixture of a lot of things like dough, sauce, meat, veggies, cheese, etc. and each of those things is a mixture of other things like proteins, starches, sugars, water, fiber, vitamins, minerals, etc.Is air a heterogeneous mixture?
A mixture in which its constituents are not distributed uniformly is called heterogeneous mixture, such as sand in water. One example of a mixture is air. Air is a homogeneous mixture of the gaseous substances nitrogen, oxygen, and smaller amounts of other substances. Mixtures can have any amounts of ingredients.What are the three types of mixtures?
Three types of mixtures based on particle size are solutions, suspensions, and colloids, all of which are described in the Table below. A solution is a homogeneous mixture with tiny particles.What is a natural emulsifier?
INCI: Cetearyl Olivate, Sorbitan Olivate. Accepted by Ecocert and most organic certifiers. This emulsifier is derived from natural olive chemistry. It is an emulsifier and thickener in one which is compatible with a wide variety of cosmetic and active ingredients over a wide pH range (3 to 12).Can you mix water and oil?
And it's not a bad analogy; oil and water won't immediately mix. Oil molecules, however, are non-polar, and they can't form hydrogen bonds. If you put oil and water in a container, the water molecules will bunch up together and the oil molecules will bunch up together, forming two distinct layers.How can you separate oil and water?
If you take a jar and fill it with water, put some 'straightforward' oil on top and shake it, all you need to do to separate the oil and the water is to put the jar on a table and wait. Shortly there will be a layer of oil floating on the surface and all that is left to do is to remove the oil layer.What happens when you mix oil and water?
So what happens when you try to mix oil and water? The water molecules attract each other, and the oil molecules stick together. That causes oil and water to form two separate layers. Water molecules pack closer together, so they sink to the bottom, leaving oil sitting on top of the water.Why is oil insoluble in water?
Many substances do not dissolve in water and that is because they are non-polar and do not interact well with water molecules. A common example is oil and water. Oil contains molecules that are non-polar, thus they do not dissolve in water.What can dissolve oil?
A variety of substances will dissolve oil, including gasoline and carbon tetrachloride -- both of which have non-polar molecules. Acetone is a special class of solvent called “dipolar aprotic” that, depending on the circumstances, can act as a weak acid or base; it dissolves oil and mixes with water as well.Can you emulsify oil and water?
If you shake the oil and water together then the oil breaks up into tiny droplets and becomes distributed in the water forming a mixture. By vigorously mixing the emulsifier with the water and fat/oil, a stable emulsion can be made. Commonly used emulsifiers include egg yolk, or mustard.Is water polar or nonpolar?
Water (H2O) is polar because of the bent shape of the molecule. The reason the shape of the molecule isn't linear and nonpolar (e.g., like CO2) is because of the difference in electronegativity between hydrogen and oxygen.Does oil and vinegar mix?
Oil and vinegar do not mix or even if they are mixed they will quickly separate when given the opportunity. Some proteins such as eggs are emulsifiers that will cause oil and vinegar to mix.Is Vinegar an emulsifier?
A good emulsifying agent will keep the oil and vinegar mixed together, delaying the appearance of a clear boundary. Try variations with more egg white, or with other emulsifiers, such as egg yolk, balsamic vinegar instead of white vinegar, etc.Is glycerin an emulsifier?
Vegetable glycerin works as an emulsifier, making it useful for blending both water-based and oil-based ingredients. It can also help thicken any blends you want to make.Is honey an emulsifier?
While honey is not an emulsifier, its thick consistency helps to stabilize the mixture.