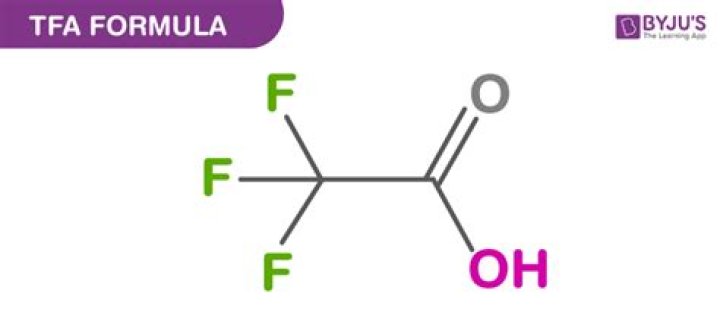
Trifluoroacetic acid (TFA) is an organofluorine compound with the chemical formula CF3CO2H. It is a structural analogue of acetic acid with all three of the acetyl group's hydrogen atoms replaced by fluorine atoms and is a colorless liquid with a vinegar like odor..
Also to know is, why TFA is used in HPLC?
TFA (trifluoroacetic acid) is a commonly used mobile phase additive for reversed-phase HPLC (RP-HPLC) separations of proteins and peptides. However, TFA interferes with and significantly reduces the LC/MS signal, lowering sensitivity.
Beside above, is TFA soluble in water? TFA is almost 100, 000-fold more acidic than acetic acid. TFA is widely used in organic chemistry. Trifluoroacetic acid appears as a colorless fuming liquid with a pungent odor. Soluble in water and denser than water.
Moreover, is TFA toxic?
Safety. Trifluoroacetic acid is a corrosive acid but it does not pose the hazards associated with hydrofluoric acid because the carbon-fluorine bond is not labile. TFA is harmful when inhaled, causes severe skin burns and is toxic for aquatic organisms even at low concentrations.
How do I remove TFA from a reaction?
Popular Answers (1) To remove traces of TFA you can use exsiccator with KOH and - optionally - some heat. If you have the salt with TFA you could dissolve your product in water add some NH3 - until you have slight alkalline conditions - and extract your product with CHCl3 or DCM, evaporate and dry over KOH.
Related Question Answers
Why acetonitrile is used in HPLC?
The B solvent is generally an HPLC grade organic solvent such as acetonitrile or methanol with 0.1% acid. The acid is used to the improve the chromatographic peak shape and to provide a source of protons in reverse phase LC/MS. In our work we use acetonitrile as our organic solvent.Why pH is important in HPLC?
As pH decreases bases gain a proton – removing H+ from the acidic environment. The reason the pKa of your analytes is so important in HPLC is because when the pH is set close to the pKa, the analytes will be present in solution in both neutral and ionized forms.What is degassing in HPLC?
Outgassing is the term used to describe dissolved gases coming out of solution. This phenomenon can also occur in a HPLC system where rough surfaces produce nucleation sites for bubble formation. In HPLC analysis the problems produced by bubble formation can largely be prevented, by degassing the mobile phase.What is USP tailing factor?
Definition: Tailing factor The tailing factor is a measure of peak tailing. It is defined as the distance from the front slope of the peak to the back slope divided by twice the distance from the center line of the peak to the front slope, with all measurements made at 5% of the maximum peak height.Why phosphate buffer is used in HPLC?
Since the retention of ionizable compounds is very sensitive to the mobile phase pH, it is necessary to control the pH of the mobile phase by the addition of a buffer. A buffer maintains the pH when a small amount of acid or base is added. The most popular buffers for HPLC with UV detection are phosphate and acetate.What is the pH of 0.1% TFA?
The pKa of TFA is in the pH 0.2-0.5 region, depending on the reference you select. 0.1% TFA gives a pH of about 1.8-2.0. This means that for pH 2.5, you would add less than 0.1% TFA. Now remember that TFA also can act as an ion-pairing reagent, as well.How does reverse phase work in HPLC?
Reversed-phase chromatography is a technique using alkyl chains covalently bonded to the stationary phase particles in order to create a hydrophobic stationary phase, which has a stronger affinity for hydrophobic or less polar compounds. Reversed-phase chromatography employs a polar (aqueous) mobile phase.What is ion pairing?
Ion pair. An ion pair, for the physicist, is the positively charged particle (positive ion) and the negatively charged particle (negative ion) simultaneously produced by the addition of sufficient energy to a neutral atom or molecule to cause it to dissociate into oppositely charged fragments.Is cf3cooh an acid or base?
The conjugate base of CF3COOH is CF3COO-, but the conjugate of the other acid is CH3OH (methanol, and NOT an anion).Is TFA volatile?
TFA as its protonated CF3COOH form is highly volatile. You need to find a stronger acid to transfer TFA to its protonated species. Its a competition reaction - the stronger acid displaces the weaker acid from its salt. Whenever TFA is protonated, if evaporates really easy from your sample.Is TFA flammable?
Trifluoroacetic Acid itself does not burn. * POISONOUS GASES ARE PRODUCED IN FIRE, including Hydrogen Fluoride. * CONTAINERS MAY EXPLODE IN FIRE. * Use water spray to keep fire-exposed containers cool.Which is stronger trichloroacetic acid or trifluoroacetic acid?
Trifluoroacetic acid [TFA] is more acidic than Trichloroacetic acid [TCA]. electronegative Cl stabilizing that negative charge (and giving a nice stable full electron orbital) vs the very non-electronegative carbon not being able to stabilize that negative charge of CH3-.