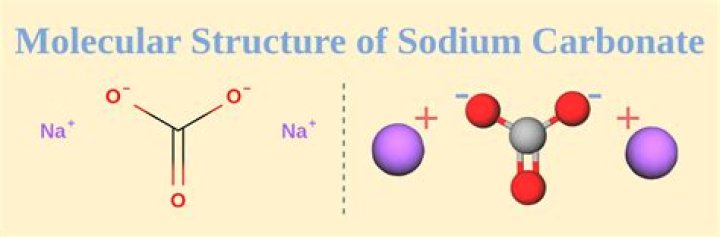
Chemical properties: Sodium carbonate is a stable but hygroscopic solid (absorbs water from air) and readily dissolves in water to form weakly acidic carbonic acid and the strong base, sodium hydroxide. Thus, the aqueous solution of Na2CO3 is overall a strong base. It reacts violently with many acids..
Just so, what is the nature of Na2CO3?
Na2CO3 is a salt of strong base(NaOH) & weak acid (H2CO3). SO,cation of strong base (Na+) can't get hydrolysed in water. so, it can't effect the the pHof water. But anion Co3 2-(carbonate ion) of weak acid get's hydrolysed in water & make solution basic.
Subsequently, question is, is sodium carbonate a base or acid? Sodium Carbonate is the disodium salt of carbonic acid with alkalinizing property. When dissolved in water, sodium carbonate forms carbonic acid and sodium hydroxide. As a strong base, sodium hydroxide neutralizes gastric acid thereby acting as an antacid.
Moreover, why is sodium carbonate basic in nature?
The aqueous solution of sodium carbonate is basic because sodium carbonate dissolves in water, it undergoes dissociation and forms three ions. These ions include two sodium and one carbonate ion.
What is the pH of sodium carbonate?
| Base | Normality | pH |
| Sodium bicarbonate (baking soda) | 0.1 N | 8.4 |
| Sodium carbonate (washing soda) | 0.1 N | 11.6 |
| Sodium hydroxide (caustic soda) | N | 14.0 |
| Sodium hydroxide | 0.1 N | 13.0 |
Related Question Answers
What is the use of Na2CO3?
The main uses of sodium carbonate are as water softener, food processing aid, pH modifier, swimming pool chemical and electrolyte. It is also used in the manufacture of glass, paper, soaps and detergents, and many other useful chemicals.How Na2CO3 is formed?
One important industrial process is the Solvay process in which sodium hydrogen carbonate (sodium bicarbonate, NaHCO3) and sodium carbonate (soda ash, Na2CO3) are produced from ammonia, carbon dioxide, water, and concentrated brine solution.Is Na2CO3 a strong or weak base?
Sodium bicarbonate is used as an anti-acid medication. Carbonates are made from reaction between carbonic acid (aqueous carbon dioxide) and a base (or alkali). They have the formula Mx(CO3)y (e.g. Na2CO3, soda ash). Carbonate salts are generally considered weak bases, and they turn litmus paper blue.Is sodium carbonate a strong or weak base?
Sodium carbonate is a basic salt which is formed by the combination of sodium hydroxide and carbonic acid. Sodium hydroxide is a strong base, while cabonic acid is a weak acid resulting in the formation of sodium carbonate salt.What is the chemical name for washing soda?
Sodium carbonate
Is HCl a strong acid?
Why is hydrochloric acid known as the strong acid? It has a very low pH, extremely acidic, higher concentration of H+ ions than OH- ions and its extent of ionisation/dissociation of H+ ions when dissolved in acid is high. HCl completely dissociates. So HCl a strong acid.Which type of salt is Na2CO3 and why?
Sodium Carbonate is the disodium salt of carbonic acid with alkalinizing property. When dissolved in water, sodium carbonate forms carbonic acid and sodium hydroxide. As a strong base, sodium hydroxide neutralizes gastric acid thereby acting as an antacid.How do you identify Na2CO3?
Using a plastic pipette, add about 2 ml of the hydrochloric acid solution into the first beaker. If bubbles of the gas (carbon dioxide) intensely evolve during the reaction, then the sample is a carbonate salt (NaHCO3 or Na2CO3); proceed to the next step.Is NaF an acid or base?
For example, NH4Br is the salt of a weak base (NH3) and a strong acid (HBr), so the salt is acidic. Similarly, NaF is basic (it is the salt of a strong base, NaOH, and a weak acid, HF). NaCl is neutral.Is NaOH an acid or base?
NaOH is a base because when dissolved in water it dissociates into Na+ and OH- ions. It is the OH- (hydroxyl ion) which makes NaOH a base. In classical term a base is defined as a compound which reacts with an acid to form salt and water as depicted by the following equation.Is AlCl3 an acid or base?
AlCl3 is acidic. But wait, now I've an acid, i.e. hydrochloric acid (HCl) and probably a base (Al(OH)3). Close enough, but Al(OH)3 is actually amphoteric in nature, which means it can behave both as an acid and a base.Is CaCl2 an acid or base?
Further, the acid (HCl) and base (NaOH) used are considered to be either a strong acid and a strong base, respectively, such that the salt will be neutral (if either/both are weak then things change). So, by analogy, CaCl2 is neither an acid or a base…just a neutral salt.Is AgNO3 an acid or base?
No, AgNO3 does not form an acidic solution nor a basic solution. In order for something to be considered an acid or base it has to produce either H+ or OH- in water. For example, HCl is an acid, one of the strongest, because it has an H+ attaches to the Cl-.Is NH4Cl an acid or base?
As mentioned in the other answer, NH4Cl is an “acidic” salt, formed by the neutralization of a strong acid (HCl) with a weak base (NH3). Therefore, when the salt is completely dissociated in an aqueous solution, it forms NH4+ and Cl- ions.Is CH3COONa an acid or base?
Sodium acetate (CH3COONa) is a salt in solid state and can't be regarded as an acid or base in anhydrous or molten form. Now, as NaOH is a strong base and CH3COOH is a weak acid, the resultant solution is basic in nature.Is carbonate a weak base?
Carbonates are made from reaction between carbonic acid (aqueous carbon dioxide) and a base (or alkali). They have the formula Mx(CO3)y (e.g. Na2CO3, soda ash). Carbonate salts are generally considered weak bases, and they turn litmus paper blue.What is the common name for sodium chloride?
Sodium chloride /ˌso?di?m ˈkl?ːra?d/, commonly known as salt (though sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions.What happens when you mix sodium carbonate and water?
When dissolved in water, sodium carbonate forms carbonic acid and sodium hydroxide. As a strong base, sodium hydroxide neutralizes gastric acid thereby acting as an antacid. Sodium carbonate is an organic sodium salt and a carbonate salt.Is sodium carbonate soluble in water?
Water