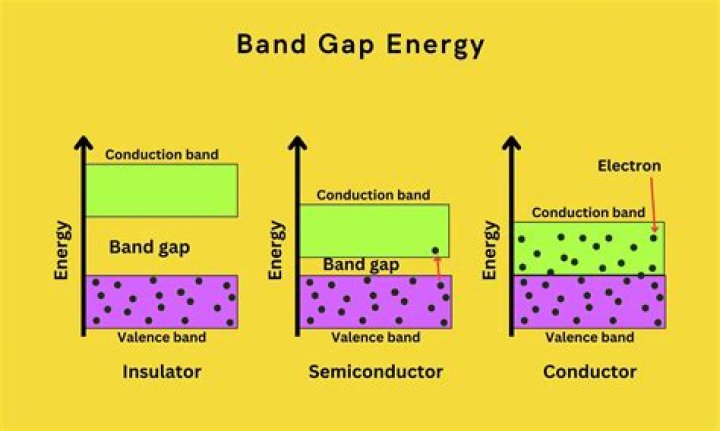
An electronic band gap is the difference in energy between the highest occupied electron state and the lowest unoccupied electron state at zero temperature. The optical band gap is the lowest energy for which a photon can be absorbed or emitted by a single photon process which conserves Crystal momentum - Wikipedia..
Besides, what is the difference between optical band gap and electronic band gap?
Electrical band gap is the minimal energy required to create an electron hole pair in a semiconductor, whereas optical band gap is the exciton energy which determines the onset of vertical interband transitions.
Similarly, what is the range of energy band gap of a semiconductor? Conventional semiconductors like silicon have a bandgap in the range of 1 - 1.5 electronvolt (eV), whereas wide-bandgap materials have bandgaps in the range of 2 - 4 eV. Generally, wide-bandgap semiconductors have electronic properties which fall in between those of conventional semiconductors and insulators.
Correspondingly, what does band gap mean?
Definition of bandgap. : the difference in energy between the valence band and the conduction band of a solid material (such as an insulator or semiconductor) that consists of the range of energy values forbidden to electrons in the material.
Why is band gap important?
Hence there is a well defined gap between the valence band and the conduction band. This is called as the 'electron band gap' and is an important concept to understand many phenomena associated with the nanomaterials. The band gap is a useful predictor of wavelength of light that will absorbed by the material.
Related Question Answers
Why does band gap decrease with temperature?
How does temperature affect the band gap? As temperature increases, the band gap energy decreases because the crystal lattice expands and the interatomic bonds are weakened. Weaker bonds means less energy is needed to break a bond and get an electron in the conduction band.Does band gap change with temperature?
The band-gap energy of semiconductors tends to decrease with increasing temperature. When temperature increases, the amplitude of atomic vibrations increase, leading to larger interatomic spacing. Band gaps also depend on pressure. Band gaps can be either direct or indirect, depending on the electronic band structure.Why band gap is formed?
Each band is formed due to the splitting of one or more atomic energy levels. Therefore, the minimum number of states in a band equals twice the number of atoms in the material. The reason for the factor of two is that every energy level can contain two electrons with opposite spin.How is a band gap formed?
When two or more atoms join together to form into a molecule, their atomic orbitals overlap. The inner electron orbitals do not overlap to a significant degree, so their bands are very narrow. Band gaps are essentially leftover ranges of energy not covered by any band, a result of the finite widths of the energy bands.How can I improve my band gap?
Band gap of semiconductor changes when foreign impurities are added. Increase or decrease in band gap will depend which element in the semiconductor is replaced by which element. For exaple if you replaces O by N, both have p-orbitals involved in bond formation.What is the unit of energy band gap?
electron volts
What is conduction band in semiconductor?
The conduction band is the band of orbitals that are high in energy and are generally empty. In reference to conductivity in semiconductors, it is the band that accepts the electrons from the valence band. The conduction band can be seen in the diagram below.What is forbidden gap?
Share. Answer. Forbidden energy gap, also known as band gap refers to the energy difference (eV) between the top of valence band and the bottom of the conduction band in materials. Current flowing through the materials is due to the electron transfer from the valence band to the conduction band.What is forbidden energy gap?
Forbidden energy gap is the gap between valence band and conduction band. In other words the energy required by an electron to jump from valence band to conduction band.What is direct band gap?
In semiconductor physics, the band gap of a semiconductor can be of two basic types, a direct band gap or an indirect band gap. The band gap is called "direct" if the crystal momentum of electrons and holes is the same in both the conduction band and the valence band; an electron can directly emit a photon.What is band gap of germanium?
Band structures of Ge. Eg= 0.66 eV.What does wide band gap mean?
WBG materials are so-called due to a relatively wide energy bandgap as compared to conventional silicon. The electronic bandgap is the energy gap between the top of the valence band and the bottom of the conduction band in solid materials.What is energy band gap theory?
Insulator Energy Bands Most solid substances are insulators, and in terms of the band theory of solids this implies that there is a large forbidden gap between the energies of the valence electrons and the energy at which the electrons can move freely through the material (the conduction band).What is the value of energy gap for silicon and germanium?
Semi-conductors : a) If the forbidden energy gap between the conduction band and valence band is small (about 1 eV) then such substances are called semiconductors. Eg : Silicon & Germanium. b) For silicon forbidden energy gap is 1.1 eV and for germanium 0.72 eV.What is energy band structure?
The energy band structure is the relationship between the energy and momentum of a carrier in a solid. For an electron in free space, the energy is proportional to the square of the momentum.What is energy gap in conductor?
The range of energy in which an electron must exist in order to participate in the conduction of electricity is known as conduction band. The difference between the valence band and conduction band is known as band gap or energy gap. In conductors, the valence band overlaps with the conduction band.Why are there no holes in metals?
Electron holes in metals. Yes, I have seen in other places that metals do not have holes because their valence and conduction bonds are usually indistinct from each other, whereas holes are left in the valence band when electrons move into the conduction band in semiconductors.Why energy band gap of silicon is more than germanium?
Silicon's valence orbital is 3p2, while germanium is 4s2. Look at the periodic table, you'll see that germanium is under silicon, and hence has an extra shell (higher principal quantum number) due to it having a higher atomic number (more electrons). Silicon's valence orbital is 3p2, while germanium is 4s2.Which element has the largest band gap?
Carbon forms very strong C-C bonds so it has a very large band gap. Thermal energy available at room temperature isn't enough to excite any electrons from the filled band to the empty band. Silicon and germanium have significantly weaker bonding between their atoms.