.
Also asked, what is the basic principle of chromatography?
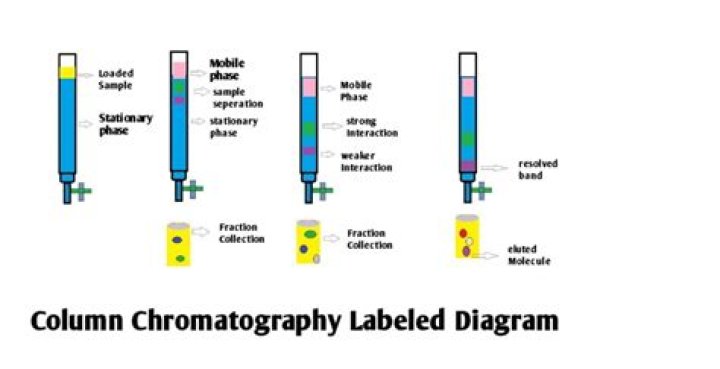
Chromatography is based on the principle where molecules in mixture applied onto the surface or into the solid, and fluid stationary phase (stable phase) is separating from each other while moving with the aid of a mobile phase.
Beside above, what is the use of column chromatography briefly explain the principle of column chromatography? Column Chromatography is a preparative technique used to purify compounds depending on their polarity or hydrophobicity. In column chromatography, a mixture of molecules is separated based on their differentials partitioning between a mobile phase and a stationary phase.
People also ask, what is column chromatography and how does it work?
Column chromatography in chemistry is a chromatography method used to isolate a single chemical compound from a mixture. Column chromatography can be done using gravity to move the solvent, or using compressed gas to push the solvent through the column.
What are the types of column chromatography?
There are three basic types of liquid chromatographic columns: liquid-liquid, liquid-solid, and ion-exchange. Liquid-liquid chromatographic columns have the liquid stationary phase bonded or absorbed to the surface of the column, or packed material.
Related Question AnswersWhy is chromatography used?
Chromatography is a method used by scientists for separating organic and inorganic compounds so that they can be analyzed and studied. Chromatography is used in many different ways. Some people use chromatography to find out what is in a solid or a liquid. It is also used to determine what unknown substances are.What is chromatography in biology?
Definition. The process or technique of separating molecules or components in a mixture according to the differential absorption and elution. Supplement. Column chromatography and paper chromatography are two of the common types of chromatography used in laboratory to separate components in a mixture.What is Rf value?
The Rf value is defined as the ratio of the distance moved by the solute (i.e. the dye or pigment under test) and the distance moved by the the solvent (known as the Solvent front) along the paper, where both distances are measured from the common Origin or Application Baseline, that is the point where the sample isWhat are the two main types of chromatography?
Chromatography utilizes phase equilibrium partitioning principles to isolate proteins, nucleic acids, or little particles in complex blends in view of their varying connections with a stationary phase and a mobile phase. There are two main types of chromatography: Liquid chromatography (LC) and gas chromatography (GC).What is an example of chromatography?
An example of chromatography is when a chemical reaction is used to cause each of the different size molecules in a liquid compound to separate into their own parts on a piece of paper. YourDictionary definition and usage example. Copyright © 2018 by LoveToKnow Corp.What is called chromatography?
Chromatography is a physical method of separation that distributes components to separate between two phases, one stationary (stationary phase), the other (the mobile phase) moving in a definite direction. The eluate is the mobile phase leaving the column. This is also called effluent.What is the origin in chromatography?
Origin. The point at which a small spot of a mixture is placed so that it can be separated by thin-layer chromatography. Retention Time. The time taken for a component to pass through a chromatography column.What is chromatography and its applications?
Chromatography is a powerful separation tool that is used in all branches of science, and is often the only means of separating components from complex mixtures. A wide range of chromatographic procedures makes use of differences in size, binding affinities, charge, and other properties.What comes out first in column chromatography?
Column chromatography can be thought of as three-dimensional version of TLC (and vice-versa). So the most polar compound which interacts with silica gel most elutes slowest and the least polar compound leaves the column first.Why Silica is used in chromatography?
Silica gel is a polar adsorbent and being slightly acidic in nature, it has a powerful capacity to absorb basic contents that may be present in the material that needs separation or purification. It is also well known for its role in reversed-phase partition chromatography.Is silica polar or nonpolar?
Silica gel, the most commonly used stationary phase, has the empirical formula SiO2. However, at the surface of the silica gel particles, the dangling oxygen atoms are bound to protons. The presence of these hydroxyl groups renders the surface of silica gel highly polar.Why Silica gel is used as stationary phase in column chromatography?
Silica and alumina are both polar adsorbents so the more polar components in the mixture to be separated are retained more strongly on the stationary phase and are therefore eluted from the column last. Typically, 70–230 silica gel is used for gravity columns and 230–400 mesh for flash columns.What solvent is used in column chromatography?
The solvent system used depends upon the behaviour of your (crude) product on silica gel. Neat hexane (or a substitute such as petroleum ether or cyclohexane) is often used to wash 'grease' (non polar compounds) off the column, whilst neat ethyl acetate (or ether) is often used to elute highly polar compounds.What is the mobile phase in column chromatography?
In column chromatography, the stationary phase, a solid adsorbent, is placed in a vertical glass (usually) column. The mobile phase, a liquid, is added to the top and flows down through the column by either gravity or external pressure.What is column efficiency?
Column efficiency, also known as plate count, is a measure of the dispersion of a peak. Narrow peaks take up less space in the chromatogram and thus allow more peaks to be separated. Efficiency is usually explained using the concept of theoretical plates.Which chemical process is stationary phase applied?
Stationary phase, in analytical chemistry, the phase over which the mobile phase passes in the technique of chromatography. Chromatography is a separation process involving two phases, one stationary and the other mobile.Do more polar solvents elute faster?
The higher the percentage of polar solvent, the faster compounds will elute. It may also be helpful to remember that alumina and silica are much more polar than any organic solvent. Therefore, the stationary phase will always be more polar than the mobile.How do you make silica gel in column chromatography?
Procedure:- Place the column in a ring stand in a vertical position.
- A plug of glass wool is pushed down to the bottom of the column.
- Prepare slurry of silica gel with a suitable solvent & pour gently into the column.
- Open the stop cock & allow some solvent to drain out.