As long as the number of protons in an atom equals the number of electrons, the atom remains uncharged, or neutral. When an atom gains or loses electrons, it becomes an electrically charged ion..
Also know, which of these relationships are true of an uncharged atom?
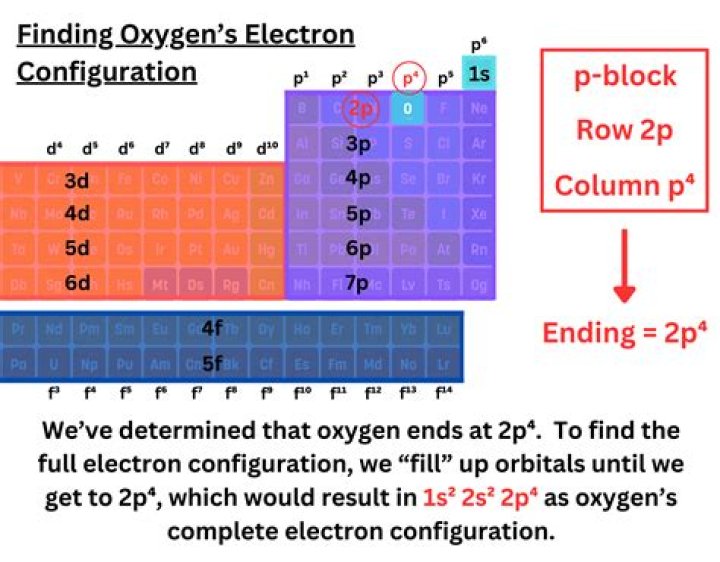
The number of protons is equal to the number of electrons. What determines the types of chemical reactions that an atom participates in? The number of electrons in the outermost electron shell.
Additionally, which statement is true of atoms? - Most of an atom's volume is filled with matter. - Protons repel electrons. - All of the above. The statement that is TRUE about atoms is “electrons determine the size of the atom.” The electrons surround the nucleus of an atom and they determine the space in which an atom occupies.
Subsequently, one may also ask, what are uncharged particles?
Neutron: An uncharged particle found in the nucleus of an atom. A neutron, like a proton, contributes one atomic mass unit to the total atomic weight of an atom. Proton: A positively charged particle found in the nucleus of an atom. This break-up gives off small fragments of atoms and energy.
How many electrons are there in an uncharged atom of element 117?
State: Uncharged atoms have the same number of electrons as protons. How many electrons are there in an uncharged atom of element 117? The element carbon has an atomic number of 6 since it has 6 protons. Its most common isotope has a mass number of 12 since it has 6 neutrons in addition to the 6 protons.
Related Question Answers
What is a trace element required by most living organisms?
Living organisms often contain trace amounts of several elements, but the most abundant ones are oxygen, carbon, hydrogen, nitrogen, calcium and phosphorus. - Oxygen. Oxygen is the most abundant element contained within living organisms, composing about 65% of the human body.
- Carbon.
- Hydrogen.
- Nitrogen.
- Sulfur.
- Phosphorus.
How are ionic bonds formed?
Ionic bond, also called electrovalent bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. Such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom.What determines the chemical behavior of an atom?
The protons and neutrons in an atom are concentrated in the atom's center. The chemical behavior of atoms is determined by the number and the configuration of the electrons in the atom.Which of these bonds is the weakest?
The weakest of the intramolecular bonds or chemical bonds is the ionic bond. next the polar covalent bond and the strongest the non polar covalent bond. There are even weaker intermolecular "bonds" or more correctly forces. These intermolecular forces bind molecules to molecules.How do u find neutrons?
Take note that the nucleus of an atom is composed of protons and neutrons. And the number of particles present in the nucleus is referred as mass number (Also, called as atomic mass). So, to determine the number of neutrons in atom, we only have to subtract the number of protons from the mass number.How do you find the atomic number?
Now you know that atomic number = number of protons, and mass number = number of protons + number of neutrons. To find the number of neutrons in an element, subtract the atomic number from the mass number.What is the atomic number of an atom that has 6 protons neutrons and electrons?
Carbon-14 (or 14C) contains six protons, eight neutrons, and six electrons; its atomic mass is 14 amu (six protons and eight neutrons).What is the difference between covalent bonds and ionic bonds?
An ionic bond is formed between a metal and a non-metal. Covalent bonding is a form of chemical bonding between two non metallic atoms which is characterized by the sharing of pairs of electrons between atoms and other covalent bonds.What is an uncharged atom called?
The Atom's Charge As long as the number of protons in an atom equals the number of electrons, the atom remains uncharged, or neutral. When an atom gains or loses electrons, it becomes an electrically charged ion. An atom that gains electrons becomes a negatively charged anion.What gives a particle charge?
Electric charge is carried by subatomic particles. In ordinary matter, negative charge is carried by electrons, and positive charge is carried by the protons in the nuclei of atoms. The proton has a charge of +e, and the electron has a charge of −e.What is a negatively charged particle?
Similarly, any particle that has a negative charge on its surface is known as a negatively charged particle. There are some fundamental particles: neutrons, protons and electrons and these make up all the atoms. A positively charged atom is called a cation and a negatively charged atom is called an anion.Can a nucleus be negatively charged?
The nucleus, that dense central core of the atom, contains both protons and neutrons. Electrons are outside the nucleus in energy levels. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. A neutral atom contains equal numbers of protons and electrons.What happens when an electron absorbs energy?
The lowest energy level an electron can occupy is called the ground state. When an electron absorbs energy, it jumps to a higher orbital. An electron in an excited state can release energy and 'fall' to a lower state. When it does, the electron releases a photon of electromagnetic energy.What happens when two atoms form a chemical bond quizlet?
An electron orbital describes a three-dimensional space where an electron can be found 90% of the time. What happens when two atoms form a chemical bond? A chemical bond forms when two atoms transfer or share outer electrons to complete their outer shells. These atoms are isotopes.Why is an atom electrically neutral?
Atoms are electrically neutral because they have equal numbers of protons (positively charged) and electrons (negatively charged). If an atom gains or loses one or more electrons, it becomes an ion.Are hydrogen bonds covalent?
Covalent bonds and hydrogen bonds are primary intermolecular forces. Covalent bonds can occur between most elements on the periodic table. Hydrogen bonds are a special bond between a hydrogen atom and an oxygen, nitrogen or fluorine atom.Who was the first to state the concept of an atom?
Democritus
What is the excited state of an atom?
An excited-state atom is an atom in which the total energy of the electrons can be lowered by transferring one or more electrons to different orbitals. That is, in an excited-state atom not all electrons are in the lowest possible energy levels. eg. Consider a carbon atom whose electron configuration is the following.