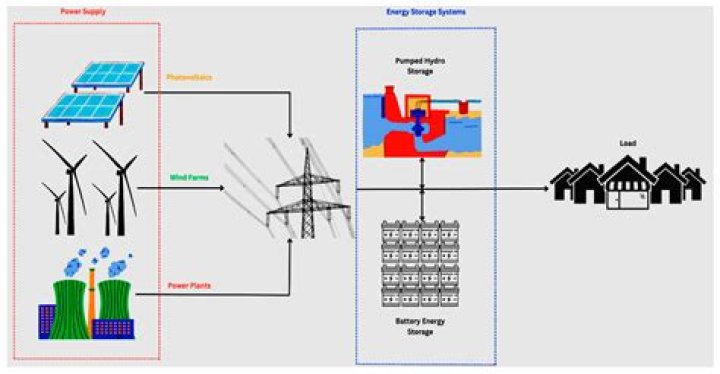
-273.15 degrees Celsius (-459.67 degrees Fahrenheit) isthe temperature at which no more energy can be removedfrom matter. It is called Absolute Zero and marks the 0 for theKelvin and Rankine scale..
Hereof, what happens when thermal energy is removed?
Surrounding matter often absorbs thermal energy.If you measured thermal energy in the matter and thesurrounding matter, you would find that energy is alsoconserved. to water vapor when thermal energy is removedfrom it. of matter that results when thermal energy is addedto a liquid.
Secondly, which phase of matter has the most potential energy? All particles have energy, but the energyvaries depending on the temperature the sample of matter isin. This in turn determines whether the substance exists in thesolid, liquid, or gaseous state. Molecules in the solidphase have the least amount of energy, while gasparticles have the greatest amount ofenergy.
Beside this, which is the transfer of thermal energy in matter with no overall transfer of matter?
Conduction is the transfer of thermal energywithout transferring matter. This device, called Newton'scradle, helps to visualize conduction. After one ball strikes therest, most of the kinetic energy is transferred toone ball on the end.
How does thermal energy produce a change from one state to another?
You can take matter from one state to another byadding or removing energy. Substances change statewhen its thermal energy increases or decreases sufficiently.Particles of a liquid have more thermal energy thanparticles of the same substance in a solid form.
Related Question Answers
What happens to a solid when energy is removed?
When a solid melts all of the energy goesinto breaking the attractions that hold the molecules in place.Because you are adding energy melt is an endothermic change.When a liquid turns into a solid the molecules slow down andbegin to vibrate in place. To cause freezing energy must beremoved.What happens when you remove heat from gas?
If you heat up a solid - you give it someenergy. This causes the particles to move more and the solid tochange into a liquid, or melt. If the liquid is heated then it mayevaporate and turn into a gas. Taking energy away from agas (cooling it down) may cause it to turn into a liquid -or condense.What is it called when heat no longer flows?
Radiation is a method of heat transfer thatdoes not require particles to carry the heat energy.Instead, heat is transferred in infrared waves (part of theelectromagnetic spectrum). Heat waves radiate out from hotobjects in all directions, travelling at the speed of light, untilthey hit another object.Do solids have potential energy?
Atoms in molecules possess potential energybecause they are attracted to each other. Potential energyleads to cohesive forces in matters, which bring particlestogether, forming liquids and solids.Does ice or water have more potential energy?
Solids have least potential energy andgases have more. Ice has lesser potentialenergy than water. Water when stored ispotential and as a freely flowing has kineticenergy associated with it.Why is boiling water a physical change?
Boiling water: Boiling water is an exampleof a physical change and not a chemical changebecause the water vapor still has the same molecularstructure as liquid water (H2O). If the bubbleswere caused by the decomposition of a molecule into a gas (such asH2O →H2 and O2), thenboiling would be a chemical change.How is thermal energy created?
Matter is made up of atoms and molecules (groupings ofatoms) and energy causes the atoms and molecules to alwaysbe in motion - either bumping into each other or vibrating back andforth. The motion of atoms and molecules creates a form ofenergy called heat or thermal energy which is presentin all matter.How can removing thermal energy affect the state of matter?
Adding or removing thermal energy from asubstance causes a change of state. Energy affectsthe attraction between the atom or molecules and their rate ofmovement. A substance's temperature determines whether it occurs ina solid, liquid or gas state.Which material is a good conductor of thermal energy?
Conduction is usually faster in certain solids andliquids than in gases. Materials that are good conductorsof thermal energy are called thermal conductors. Metalsare especially good thermal conductors because they havefreely moving electrons that can transfer thermal energyquickly and easily.What is the transfer of energy by waves moVing through space?
RADIATION is the transfer of energy as wavesmoVing through space.Which type of thermal energy uses electromagnetic waves to transfer energy Brainly?
The correct answer is radiation thermal energy.Explanation: The transfer of energy in thermalenergy comprises the transfer of internal energy.The three kinds of thermal energy transfer are convection,conduction, and radiation.Why is conduction slower in gases than in liquids and solids?
Conduction is the transfer of thermal energy withno transfer of manner. Conduction in gas is slowerthan in liquids and solids because the particles in agas collide less often. Why is conduction slower in gasesthan in liquids or solids? Conduction in metalsis faster because the electrons are free to moveabout.What is the highest potential energy?
Explanation: Potential energy is theenergy a body has due to its position rather than itsmotion, which would be kinetic energy. The order ofdecreasing potential energy for a substance is solid <liquid < gas < plasma. The solid state would have thegreatest potential energy and least kineticenergy.What is the fifth state of matter?
In 1924, Albert Einstein and Satyendra Nath Bosepredicted the "Bose-Einstein condensate" (BEC), whichis referred as the fifth state of matter. A Bose-Einsteincondensate (BEC) is a state of matter of a dilute gas ofbosons cooled to temperatures very close to absolute zero (that is,very near 0 K or ?273.14 C).What are the 12 states of matter?
There are four natural states of matter: Solids,liquids, gases and plasma. The fifth state is the man-madeBose-Einstein condensates. In a solid, particles are packed tightlytogether so they don't move much.How do scientists use the kinetic theory of matter?
Scientists think that the particles of allmatter are in constant motion. The theory that allmatter consists of constantly moving particles is called thekinetic theory of matter. If particles do not haveenough kinetic energy to overcome the force of attractionbetween them, matter exists as a solid.Why does matter have potential energy?
All matter also has potential energy, orenergy that is stored and can be released at a later time.The energy of motion is called It depends on both anobject's mass and the speed at which the object is moving. The morematter an object contains, the greater itsmass.What is kinetic theory of matter?
The Kinetic Theory of Matter states thatmatter is composed of a large number of smallparticles—individual atoms or molecules—that are inconstant motion. This theory is also called theKinetic-Molecular Theory of Matter and the KineticTheory of Gases.What state of matter has the most energy?
The gas state of matter has the most energybecause of how freely the molecules move. Matter is a physicalsubstance of which there are three, and they are solid, liquid, andgas. Solid matter is very compact with the particles veryclose together and not much movement; therefore, no action, noenergy.