To calculate bond order in chemistry, subtract the number of the electrons in the antibonding molecules from the number of electrons in the bonding molecules. Divide the result by 2 to get the result. The higher the bond order, the more stable the molecule.
.
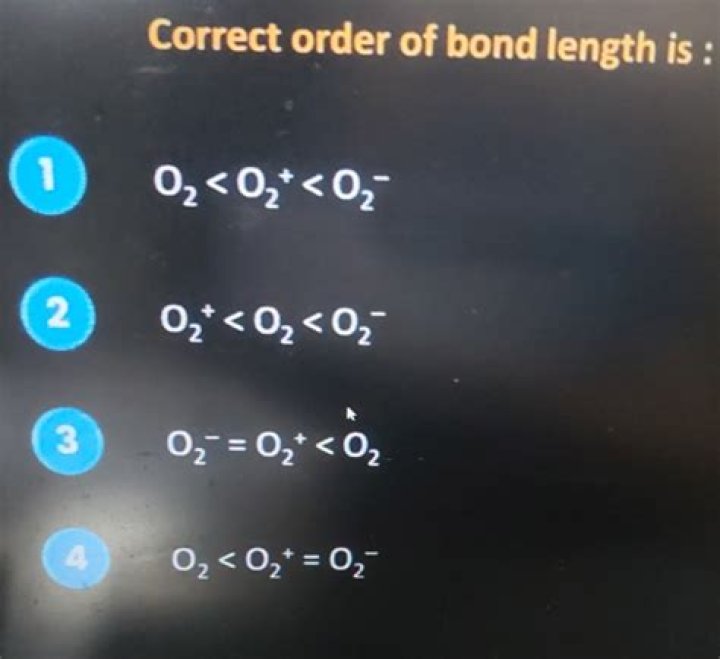
In this way, which has highest bond order between o2 and o2+?
O2+ has more bond dissociation energy than O2. Because the bond order in O2 is 2 where as in O2+ is 2.5.
Also Know, why is the bond order of o2 less than o2+? explain why bond order of N2 is greater than N2+ but the bond order of O2 is less than O2+ Bond order = No. of electron in bonding MOs - No. of electron in antibonding orbital/2.
Similarly, what is the bond order for o2+?
There are 10 electrons in BMO and 5 electrons in ABMO . So the bond order of O2+ is 2.5.
What is the bond order of CN?
The bond order of CN- is 3. Since all the electrons are paired, this ion should be diamagnetic.
Related Question AnswersWhy does o2 have a bond order of 2?
In normal O2, there are 6 bonding electrons and 2 antibonding electrons, making the bond order 2. By removing the 2 highest electrons, which reside in antibonding orbitals, to make O2^(2+), the calculation becomes (6–0)/2 = 3.Is o2+ more stable than o2?
O2+ is more stable than O2-. Reason: According to molecular orbital theory O2+ has 15 electrons &it has one electron in antibonding orbital. In the case of O2- 17 electrons are present &3 electrons are present in antibonding orbitals. If number of electrons more in antibonding orbital the molecule become unstable.What is the bond dissociation energy for CC?
It takes roughly 100 kcal of energy to break 1 mol of C–H bonds, so we speak of the bond energy of a C–H bond as being about 100 kcal/mol. A C–C bond has an approximate bond energy of 80 kcal/mol, while a C=C has a bond energy of about 145 kcal/mol.What is the bond length of o2?
Molecular Parameters - Oxygen - Selenium Compounds| Molecular Formula | Name | Bond Length (Å) |
|---|---|---|
| O2 | oxygen | O-O 1.208 |
| [O2]+ | O-O 1.1227 | |
| [O2]-(s) | O-O 1.28 (s=0.01) | |
| [O2]2-(s) | O-O 1.49 (s=0.009) |
How do you find the bond order?
If there are more than two atoms in the molecule, follow these steps to determine the bond order:- Draw the Lewis structure.
- Count the total number of bonds.
- Count the number of bond groups between individual atoms.
- Divide the number of bonds between atoms by the total number of bond groups in the molecule.