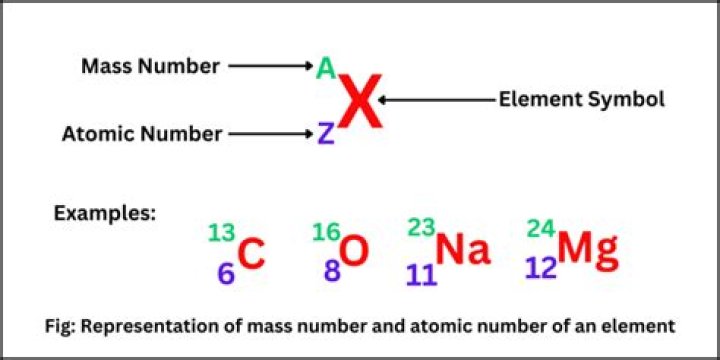
For any given isotope, the sum of the numbers of protons and neutrons in the nucleus is called the mass number. This is because each proton and each neutron weigh one atomic mass unit (amu). By adding together the number of protons and neutrons and multiplying by 1 amu, you can calculate the mass of the atom..
Keeping this in view, what 3 types of information are needed to calculate an average atomic mass?
The Number Of Isotopes That Exist For The Element. The Atomic Number For The Element. The Percentage Abundance Of Each Isotope.
One may also ask, where can you go to find information about the atomic mass of an element? Most naturally-occurring elements consist of mixtures of isotopes, so you can't just add the masses of the protons and neutrons in a single atom. Look up the atomic mass in the Periodic Table. For example, the atomic mass of Cl is 35.45 u. Take the weighted average of the atomic masses of the element's isotopes.
Just so, why is it necessary to calculate the average atomic mass for an element?
Why is it necessary to use the average atomic Mass of all isotopes, rather than the mass of the most commonly occurring isotope, when referring to the atomic mass of an element? Using a weighted average atomic mass, you can account for the less common isotopes.
What is the formula for atomic mass?
Calculating Average Atomic Mass Average atomic mass = f1M1 + f2M2 +… + fnMn where f is the fraction representing the natural abundance of the isotope and M is the mass number (weight) of the isotope. The average atomic mass of an element can be found on the periodic table, typically under the elemental symbol.
Related Question Answers
What has a mass of 1 amu?
An atomic mass unit (symbolized AMU or amu) is defined as precisely 1/12 the mass of an atom of carbon-12. The carbon-12 (C-12) atom has six protons and six neutrons in its nucleus. In imprecise terms, one AMU is the average of the proton rest mass and the neutron rest mass.What is atomic mass number?
The mass number (symbol A, from the German word Atomgewicht [atomic weight]), also called atomic mass number or nucleon number, is the total number of protons and neutrons (together known as nucleons) in an atomic nucleus. The mass number is different for each different isotope of a chemical element.How can I calculate average?
How to Calculate Average. The average of a set of numbers is simply the sum of the numbers divided by the total number of values in the set. For example, suppose we want the average of 24 , 55 , 17 , 87 and 100 . Simply find the sum of the numbers: 24 + 55 + 17 + 87 + 100 = 283 and divide by 5 to get 56.6 .How do you find Amu average?
The average atomic mass for an element is calculated by summing the masses of the element's isotopes, each multiplied by its natural abundance on Earth. When doing any mass calculations involving elements or compounds, always use average atomic mass, which can be found on the periodic table.How do you find the electrons?
The number of protons, neutrons, and electrons in an atom can be determined from a set of simple rules. - The number of protons in the nucleus of the atom is equal to the atomic number (Z).
- The number of electrons in a neutral atom is equal to the number of protons.
What is the atomic mass of an element quizlet?
AMU is defined as 1/12th of the mass of a carbon-12 atom. This is the isotope chosen to be used as a reference standard to compare atomic mass among atoms. What is the Atomic Mass of an element? The atomic mass of an element is the weighted average mass of the atoms in a naturally occurring sample of the element.How do you find the empirical formula?
What is the empirical formula of the compound? Start with the number of grams of each element, given in the problem. Convert the mass of each element to moles using the molar mass from the periodic table. Divide each mole value by the smallest number of moles calculated.Whats is an atom?
An atom a fundamental piece of matter. An atom itself is made up of three tiny kinds of particles called subatomic particles: protons, neutrons, and electrons. The protons and the neutrons make up the center of the atom called the nucleus and the electrons fly around above the nucleus in a small cloud.What is in a mole?
A mole is the atomic weight of a molecule of the chemical in grams. So a mole of a molecule like hydrogen (H) with an atomic weight of 1 is one gram. But even though the weight is different, the two moles contain the exact same number of molecules, 6.02 x 10 to the 23rd power.How do you calculate the atomic mass of oxygen?
15.999 u
How do you calculate mass number?
Together, the number of protons and the number of neutrons determine an element's mass number: mass number = protons + neutrons. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number.What is the equation for atomic number?
Atomic Number and Atomic Weight. Atomic number: Number of protons present in the nucleus is called atomic number (Z) of an element. Atomic weight: The sum of mass number of proton and mass number of neutron is called atomic weight or atomic mass number (A). Z = 19 and A = 39 as per atomic weight definition.How do you solve isotopes?
The mass number of an isotope represents the mass of the isotope's protons and neutrons. Calculate the number of neutrons in an isotope, by subtracting the atomic number from the mass number. For example, carbon-12 has six neutrons, since the atomic number of carbon is six.How do you find the amount of neutrons?
Take note that the nucleus of an atom is composed of protons and neutrons. And the number of particles present in the nucleus is referred as mass number (Also, called as atomic mass). So, to determine the number of neutrons in atom, we only have to subtract the number of protons from the mass number.Is atomic mass and mass number the same?
Atomic mass is the weighted average mass of an atom of an element based on the relative natural abundance of that element's isotopes. The mass number is a count of the total number of protons and neutrons in an atom's nucleus.Why is the atomic mass a decimal?
Though individual atoms always have an integer number of atomic mass units, the atomic mass on the periodic table is stated as a decimal number because it is an average of the various isotopes of an element.How do you find the molecular formula?
Divide the molar mass of the compound by the empirical formula molar mass. The result should be a whole number or very close to a whole number. Multiply all the subscripts in the empirical formula by the whole number found in step 2. The result is the molecular formula.Who Discovered Iron?
In Mesopotamia (Iraq) there is evidence people were smelting iron around 5000 BC. Artifacts made of smelted iron have been found dating from about 3000 BC in Egypt and Mesopotamia. In those times, iron was a ceremonial metal; it was too expensive to be used in everyday life.