For 4d and 5d metals (d4-d7): In general, the size of Do is greater than for 3d metals. As a result, complexes are typically low spin. Even a ligand such as chloride (quite weak) produces a large enough value of Do in the complex RuCl62- to produce a low spin, t2g4 configuration..
Herein, what are low and high spin complexes?
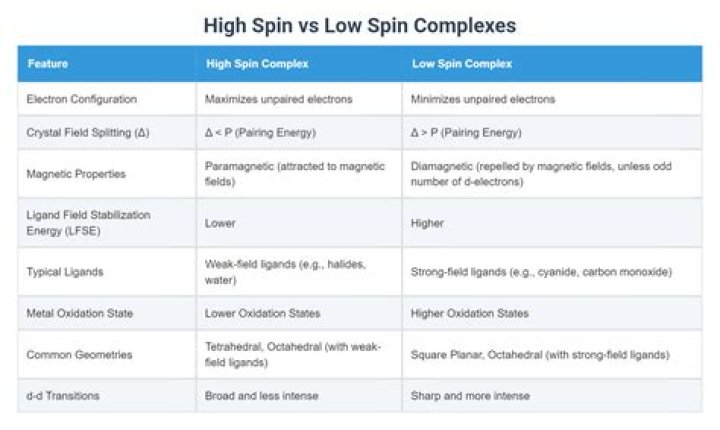
Octahedral complexes If the energy required to pair two electrons is greater than the energy cost of placing an electron in an eg, Δ, high spin splitting occurs. Complexes such as this are called "low-spin" since filling an orbital matches electrons and reduces the total electron spin.
Similarly, what makes a ligand strong or weak? Strong ligands form inner orbital and low spin complex as they help in the pairing of electrons , whereas weak ligands form outer orbital high spin complex. The weak field and strong field ligands are based on their capability of splitting.
Thereof, why are low spin tetrahedral complexes rare?
Answer: In tetrahedral complex, the d-orbital is splitting to small as compared to octahedral. Hence, the orbital splitting energies are not enough to force pairing. As a result, low spin configurations are rarely observed in tetrahedral complexes.
Why are tetrahedral complexes always high spin?
It is rare for the Δt of tetrahedral complexes to exceed the pairing energy. Usually, electrons will move up to the higher energy orbitals rather than pair. Because of this, most tetrahedral complexes are high spin.
Related Question Answers
How do you know if you have a low spin complex?
In an octahedral complex, when Δ is large (strong field ligand), the electrons will first fill the lower energy d orbitals before any electrons are placed on the higher energy d orbitals. It is then classified as low spin because there is a minimal amount of unpaired electrons.What is the meaning of low spin complex?
Definition of Low Spin Complex. Crystal field designation for an inner orbital complex; contains electrons paired t2g orbitals before eg orbitals are occupied in octahedral complexes.Is Cobalt II high spin or low spin?
As cobalt is in the +2 oxidation state, it would have a d7 configuration. Thus, if the complex is high spin, the electronic configuration is (t2g)5 (eg)2 and (t2g)6 (eg)1 if it is low spin.Is nh3 high or low spin?
For biological ligands, H2O and NH3, the most stable spin state is high spin (S ) 3/2). The difference in energy between high and low spin is dependent upon the ligand mix and coordination stereochemistry. High spin optimized geometries for these model compounds give structures close to octahedral symmetry.Is tetrahedral always high spin?
Tetrahedral Geometry Finally, the bond angle between the ligands is 109.5o. It is rare for the Δt of tetrahedral complexes to exceed the pairing energy. Usually, electrons will move up to the higher energy orbitals rather than pair. Because of this, most tetrahedral complexes are high spin.Which is strongest ligand?
CO
Is SCN a strong field ligand?
NCS- is a strong field ligand while SCN- is a weak field ligand. A spectrochemical series is the arrangement of common ligands in the increasing order of their crystal-field splitting energy (CFSE) values.Why TD complexes are mostly high spin complexes rather low spin?
It is rare for the Δt of tetrahedral complexes to exceed the pairing energy. Usually, electrons will move up to the higher energy orbitals rather than pair. Because of this, most tetrahedral complexes are high spin.What are low spin tetrahedral complexes not formed?
Now the low spin complexes are formed when a strong field ligands forms a bond with the metal or metal ion. The strong field ligands invariably cause pairing of electron and thus it makes some in most cases the last d-orbital empty and thus tetrahedral is not formed.Why square planar complexes are more stable than octahedral complexes?
These complexes differ from the octahedral complexes in that the orbital levels are raised in energy due to the interference with electrons from ligands. This means that most square planar complexes are low spin, strong field ligands.Why tetrahedral complexes do not show geometrical isomerism?
Tetrahedral complexes do not show geometrical isomerism because the relative positions of the unidentate ligands attached to the central metal atom are the some with respect to each other.Why are some complexes square planar?
The reason that many d8 complexes are square-planar is the very large amount of crystal field stabilization that this geometry produces with this number of electrons. Square planar CFT splitting: Electron diagram for square planer d subshell splitting.Why are pi complexes known as transition metals?
Transition metal form complexes due to availability of vacant d-orbitals so they can form complex with ligands. The transition metals form Π complexes because they have t2g orbitals of correct symmetry which allows them to overlap with the vacant Π orbitals of these ligands, forming Π bonds.What is the shape of Ni Co 4?
One 4s- and three 4p- orbitals undergo sp³ hybridisation and form four hybrid orbitals of equal energy which are directed towards the four corners of a regular tetrahedron. These orbitals overlap with orbitals of CO ligands. Thus, a tetrahedral [Ni(CO)4] complex is formed.Is EDTA a strong ligand?
EDTA is a moderately strong field, while (en ) is a strong field ligand.Is h2o a weak or strong field ligand?
H2O comes under the category of intermediate field ligand because it can donate only through sigma bonding. However it acts as strong ligand when metal ion is in +3 oxidation for metals like cobalt , nickel and copper and weak ligand for metals like Sc , Ti, V, Cr, Mn and Fe in +3 Oxidation state.Is oxygen a strong or weak field ligand?
Being lewis bases(those who donate electrons) the ligands with less electronegativity will be stronger. Therefore, in general halogen or oxygen donors (eg-F-,Cl-,Br-,H2O)are weak field ligands and the ones in which carbon or nitrogen atom is the donor (eg-CN-,CO,NH3) are strong field ligands.What makes a good ligand?
1 Answer. One definite factor is the ability of the ligand to stabilize its lone pair. You observe that the cyanide ion is a better ligand than the nitrogen ligand. The lone pair that reacts on the cyanide ion is the lone pair on the carbon atom.How do you know if a complex is octahedral or tetrahedral?
How do we tell whether a particular complex is octahedral, tetrahedral, or square planar? Generally speaking, octahedral complexes will be favored over tetrahedral ones because: It is more (energetically) favorable to form six bonds rather than four. The CFSE is usually greater for octahedral than tetrahedral complexes