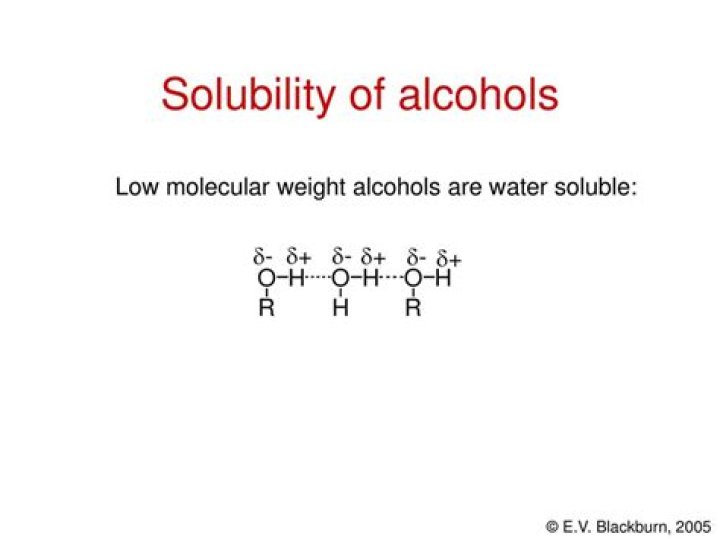
Answer : The low molecular mass alcohols are soluble in water due to presence of intermolecular hydrogen bonding between alcohol molecules and this is possible due to the presence of polar –OH group in alcohol. … Thus, solubility of alcohol decreases with increases in molecular size.
Are low molecular weight alcohols soluble in water?
Alcohols of low molecular weight are soluble in water while that of high molecular weight are insoluble in water . because of high hydrogen bonding in low molecular weight Alcinous.
Why alcohols of lower molecular weight are more soluble in water than those of higher molecular weight?
Alcohols of four or fewer carbon atoms are soluble in water because the alcohol molecules engage in hydrogen bonding with water molecules; comparable alkane molecules cannot engage in hydrogen bonding.
Why the low molecular weight alcohols are more soluble in water than the low molecular weight ethers?
Alcohols have higher boiling point than ethers of same molecular mass because in alcohol there is the presence of intermolecular hydrogen bonding. Alcohols form H-bonds with water due to the presence of −OH group. … As a result, alcohols are comparatively more soluble in water than ethers of comparable molecular masses.Why small alcohols are soluble in water but large alcohols are not?
Lower alcohols have less carbon atoms than that of higher alcohols. Higher alcohols have large no. of hydrocarbon chains which results in more steric hindrance to make bonds which result in less solubility.
What is a low molecular weight alcohol?
Alcohols of low molecular weight are soluble in water while that of high molecular weight are insoluble in water .because of high hydrogen bonding in low molecular weight Alcinous.
Why do alcohols decrease in solubility?
Alcohols are soluble in water. … The reason why the solubility decreases as the length of hydrocarbon chain increases is because it is requires more energy to overcome the hydrogen bonds between the alcohol molecules as the molecules are more tightly packed together as the size and mass increases.
Why alcohol is more soluble in water than ethers?
In ethyl alcohol, the hydrogen atom is bonded to an electronegative oxygen atom. Thus, hydrogen bonding is possible in ethyl alcohol. … Thus, hydrogen bonding is not possible in dimethyl ether. Thus, ethyl alcohol is more soluble in water than dimethyl ether because alcohol contains hydrogen bonding.What is the reason for alcohols to be more soluble in water than ether?
The difference between the ether group and the alcohol group, however, is that the alcohol group is both a hydrogen bond donor and acceptor. The result is that the alcohol is able to form more energetically favorable interactions with the solvent compared to the ether, and the alcohol is therefore more soluble.
Why alcohol is soluble in water but ether is not?Ethers are nonpolar and can only form London forces, a very weak type of intermolecular force. So ethers cannot dissolve in water because the two types on bonding do not do well together. Alcohol also form hydrogen bonds. However, alcohols are made of a hydroxy group and an alkyl group.
Article first time published onWould you expect greater solubility in water or in hexane?
Greater solubility in hexane: toluene and isobutene. Greater solubility in water: sucrose and glycol. Polar solutes are more soluble in polar solvents (i.e., water), and nonpolar solutes are more soluble in nonpolar solvents (i.e., hexane). This is the basis of the rule of thumb that “like dissolves like.”
Which molecule would you expect to be more soluble in water ccl4 ch3cl?
Therefore, CH2Cl2 is more soluble in water than CCl4.
Why are only some alcohols soluble in water?
Explanation: Small alcohols have attached OH groups which make the alcohols polar. The polarity of the alcohol and polarity of the water create intermolecular forces, most notably dipole-dipole forces.
Why are some alcohols more soluble in water?
Because alcohols form hydrogen bonds with water, they tend to be relatively soluble in water. The hydroxyl group is referred to as a hydrophilic (“water-loving”) group, because it forms hydrogen bonds with water and enhances the solubility of an alcohol in water.
Why are alcohols more soluble in water than the hydrocarbons of comparable molecular masses?
Due to presence of –OH group, alcohols can form hydrogen bonds with water but hydrocarbons cannot form such hydrogen bonds due to the absence of –OH group. Hence, alcohols are comparatively more soluble in water than hydrocarbons of comparable molecular masses.
Why solubility of alkanes decrease with increase in molecular mass?
The solubility of a homologous series of organic compounds in water decreases with increasing chain length because water molecules become increasingly less able to participate in the strong dispersion interactions between the long-chained organic molecules.
Why is ethanol more soluble in water than hexanol?
Ethanol has an OH group and only 2 carbon atoms; 1-hexanol has one OH group for 6 carbon atoms and is thus more like a (nonpolar) hydrocarbon than ethanol is.
How do the structure and number of carbons affect the solubility of alcohols in water?
The number of carbon atoms in an alcohol affects its solubility in water, as shown in Table 13.3. As the length of the carbon chain increases, the polar OH group becomes an ever smaller part of the molecule, and the molecule becomes more like a hydrocarbon. The solubility of the alcohol decreases correspondingly.
Which one of the following alcohols is least soluble in water?
Of the given options, the largest alcohol of all is 1- pentanol and will thus have the least solubility in water. Thus, the correct answer is D.
What is the difference between high molecular weight and low molecular weight?
Lower molecular weight will typically flow easier. A High molecular weight increases the impact resistance of the material. … A High molecular weight increases the chemical resistance – to a point. It takes more damage to the main chains of the molecules before it will affect the strength of the material.
Why are phenols more acidic than alcohols?
htm. Phenols are much more acidic than alcohols because the negative charge in the phenoxide ion is not localized on the oxygen atom, as it is in an alkoxide ion, but is delocalized-it is shared by a number of carbon atoms in the benzene ring.
Why are alcohols more soluble in water than alkanes?
Alcohols of four or fewer carbon atoms are soluble in water because the alcohol molecules engage in hydrogen bonding with water molecules; comparable alkane molecules cannot engage in hydrogen bonding.
Is molecular weight of alcohol is higher than water?
Common Nameethyl alcoholAlcoholCH3CH2OHSolubility in Water, g/100 mlmiscibleB.P. (°)78.5
Why is water more polar than alcohol?
Water is much more polar than an alcohol because the latter has an organic part and a non-organic part (OH) to the molecule. That is why when the alkyl group of an alcohol gets larger it becomes more likely to dissolve organic substances (and the opposite applies.
Why ethyl alcohol is completely soluble in water?
Water is a polar solvent and has hydrogen bonding , so for any other compund to dissolve in water , it must posses Polarity or H-Bond. Ethyl alcohol shows presence of hydrogen bonding as well as it is polar due to high EN of oxygen , due to this it is soluble in water.
Which alcohols are more soluble in water?
NameFormulaSolubilityMethanolCH3OHmiscibleEthanolC2H5OHmisciblePropanolC3H7OHmiscibleButanolC4H9OH0.11
Why is ethanol soluble in water?
Ethanol is soluble in water primarily because of the presence of -OH group that allows or enables it to form hydrogen bonds with water molecules. In other words, ethanol is soluble in water because it is a polar solvent.
Why is ethanol more soluble in water than Methoxymethane?
Since ethanol has the O-H group, it is a polar molecule, and is miscible with the polar solvent water. Methoxymethane (I think that was a typo, methoxyethane isn’t an isomer of ethanol) isn’t polar, so it’s not miscible with water.
Why ethanol is soluble in water but diethyl ether not?
Explain. Alcohols can form intermolecular hydrogen bonding with water molecules while ethers cannot. Hence, ethyl alcohol is soluble in water.
Why ethanol is more soluble than methanol?
For alcohols, it is known that as the number of carbon chain or skeleton increases the lesser its solubility in that polar solvent. Ethanol has one extra carbon in its skeleton than methanol. Hence, methanol will be more soluble in water than ethanol. Methanol is more soluble.
Which molecule would you expect to be more soluble in water?
of an extra OH group allows for more hydrogen bonding with water. Hence, HOCH2CH2CH2OH can be expected to be more soluble in water.