.
Correspondingly, why are carboxylic acids important?
Carboxylic acids are also important in the manufacture of greases, crayons, and plastics . Compounds with carboxyl groups are relatively easily converted to compounds called esters, which have the hydrogen atom of the carboxyl group replaced with a group containing carbon and hydrogen atoms .
Secondly, what is carboxyl group? In chemistry, the carboxyl group is an organic, functional group consisting of a carbon atom that's double-bonded to an oxygen atom and singly bonded to a hydroxyl group. Another way to view it is as a carbonyl group (C=O) that has a hydroxyl group (O-H) attached to the carbon atom.
In respect to this, what is an example of a carboxylic acid?
A carboxylic acid is an organic compound that contains a carboxyl group (C(=O)OH). Important examples include the amino acids and acetic acid. Deprotonation of a carboxyl group gives a carboxylate anion.
What are the uses of alkanoic acid?
The following points will state other significant uses of carboxylic acids:
- Manufacturing of soaps need higher fatty acids.
- Food industry uses many organic acids for the production of soft drinks, food products etc.
- In pharmaceutical industry organic acids are used in many drugs such as aspirin, phenacetin etc.
What foods contain carboxylic acids?
Many carboxylic acids occur naturally in plants and animals. Citrus fruits such as oranges and lemons contain citric acid. Figure 25.12.2: Citric acid is a large carboxylic acid with three ionizable hydrogen atoms. It is found in citrus fruits and gives them their sour or tart flavor.Is COOH an acid or base?
Carboxyl groups are weak acids, dissociating partially to release hydrogen ions. The carboxyl group (symbolized as COOH) has both a carbonyl and a hydroxyl group attached to the same carbon atom, resulting in new properties.What are the properties of carboxylic acids?
Properties of carboxylic acids. Carboxylic acids are soluble in water. Carboxylic acids do not dimerise in water, but forms hydrogen bonds with water. Carboxylic acids are polar and due to the presence of the hydroxyl in the carboxyl group, they are able to form hydrogen bonds with water molecules.How are esters formed?
Esters are formed by the condensation reaction between an alcohol and a carboxylic acid. This is known as esterification. In a condensation reaction, two molecules join and produce a larger molecule whilst eliminating a small molecule. During esterification this small molecule is water.How is a carboxylic acid formed?
15.3: The Formation of Carboxylic Acids. As we noted previously, the oxidation of aldehydes or primary alcohols forms carboxylic acids: In the presence of an oxidizing agent, ethanol is oxidized to acetaldehyde, which is then oxidized to acetic acid. Acetic acid can be further oxidized to carbon dioxide and water.How do you name carboxylic acids?
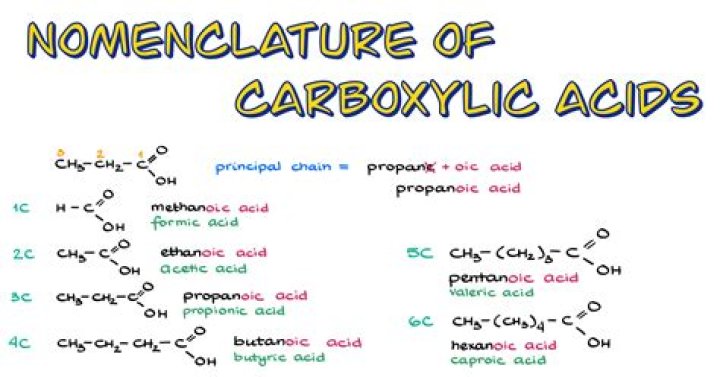
In general, carboxylic acids are named based on the number of carbons in the longest continuous chain, including the carboxyl group (-COOH). The suffix of this carbon chain is then replaced, as carboxylic acids always end in "-oic acid." An example is CH2O2, in which the longest continuous carbon chain is a methane.How do you identify carboxylic acids?
The following tests can be used to identify carboxylic acids:- Litmus Test. Carboxylic acid turns blue litmus red.
- Sodium Hydrogen Carbonate Test. Carboxylic acids reacts with sodium hydrogen carbonate to produce carbon dioxide gas which can be seen in the form of a brisk effervescence.
- Ester Test.
Why is COOH acidic?
The Carboxylic Acid naming occurs when a substance donates a proton; usually hydrogen to other things. The carboxylic acids are acidic in nature because hydrogen belongs in the -COOH group.Is Vinegar a carboxylic acid?
Vinegar is no less than 4% acetic acid by volume, making acetic acid the main component of vinegar apart from water. Acetic acid is the second simplest carboxylic acid (after formic acid). It consists of a methyl group attached to a carboxyl group.What is the name of COOH?
Originally Answered: What's the IUPAC name of cooh-cooh? IUPAC name is ethanedioic acid while common name is oxalic acid.How many types of carboxylic acids are there?
The first four carboxylic acids derived from alkanes are methanoic acid (HCOOH), ethanoic acid (CH3COOH), propanoic acid (C2H5COOH) and butanoic acid (C3H7COOH).5. What are carboxylic acids?
| Carbon atoms | 4 |
|---|---|
| Common name | Butyric acid |
| IUPAC name | Butanoic acid |
| Chemical formula | CH 3(CH 2) 2COOH |
| Common location or use | Rancid butter |